- AI
- Molecular Imaging
- CT
- X-Ray
- Ultrasound
- MRI
- Facility Management
- Mammography
Emerging Prostate and Brain MRI AI Platform Gets FDA Nod
In addition to a previously FDA-cleared brain magnetic resonance imaging (MRI) module, the Advantis Platform features an artificial intelligence-powered prostate MRI module that offers simultaneous viewing of conventional and advanced sequences, and reportedly generates automated PI-RADS assessments.
The Food and Drug Administration (FDA) has reportedly granted 510(k) clearance to the Advantis Platform (Advantis Medical Imaging), which offers artificial intelligence-enabled modules for multiparametric prostate MRI as well as brain MRI analysis.
The Advantis Prostate module streamlines radiology workflows with automated diffusion-weighted imaging (DWI) and dynamic contrast-enhanced (DCE) MRI maps, and simultaneous viewing of advanced and conventional sequences in one viewer, according to Advantis Medical Imaging. The company adds that the Advantis Prostate module provides automated PI-RADS reporting with one click.
Images courtesy of Advantis Medical Imaging.
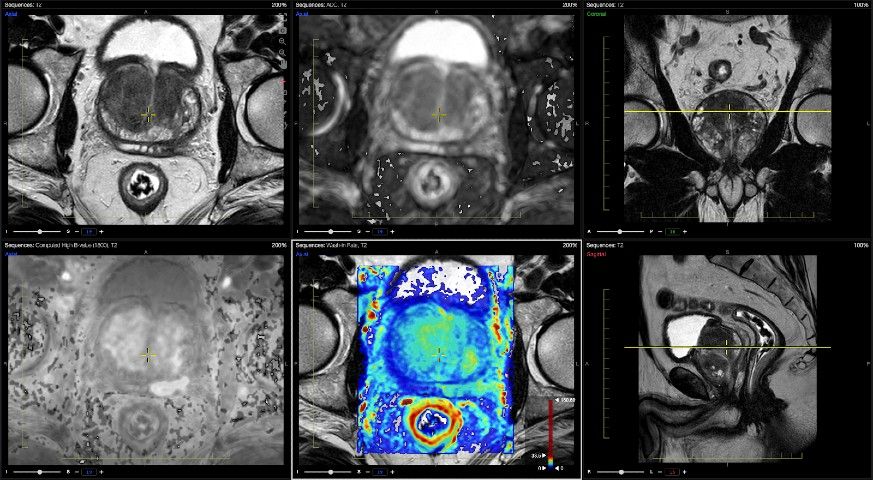
Advantis Medical Imaging said the Advantis Brain module (which previously garnered FDA 510(k) clearance under the name Brainance MD) offers a variety of features including automated processing of dynamic susceptibility contrast (DSC) MRI perfusion scans with expedient exporting of maps and key findings to the existing PACS system. Another benefit of Advantis Brain is fully automated tractography with single-click fiber tracking on diffusion tensor imaging (DTI), according to the company.
“This latest FDA clearance is a testament to our commitment to delivering innovative solutions that streamline workflows and provide clinicians with reliable, accessible and efficient software tools,” noted Zoi Giavri, the chief product office for Advantis Medical Imaging. “Our recent integration of the multiparametric prostate MRI module in our software suite, Advantis Platform, complements the existing brain MRI module while both of them enable healthcare professions to more easily extract meaningful information from MRIs.”
(Editor’s note: For related content, see “Can Explainable AI Enhance Diagnosis and PI-RADS Classification of Prostate Cancer on MRI?” and “FDA Clears New AI Software for MRI Detection of Prostate Cancer.”
ACR Collaborative Model Achieves 20 Percent Improvement in PI-QUAL Scores for Prostate MRI
May 9th 2024Using a learning network model to discuss challenges and share insights among radiology departments from five different organizations, researchers noted that 87 percent of audited prostate MRI exams had PI-QUAL scores > 4 at the conclusion of the collaborative program.
MRI-Based Deep Learning Algorithm Shows Comparable Detection of csPCa to Radiologists
May 8th 2024In a study involving over 1,000 visible prostate lesions on biparametric MRI, a deep learning algorithm detected 96 percent of clinically significant prostate cancer (csPCa) in comparison to a 98 percent detection rate for an expert genitourinary radiologist.
Emerging Perspectives on PSMA PET Radiotracers: An Interview with Kenneth J. Pienta, MD
April 24th 2024In a recent interview, Kenneth J. Pienta, M.D., discussed the impact of piflufolastat F18, current directions in research with other PSMA-targeted radiotracers and future possibilities for the role of PSMA PET in the imaging paradigm for prostate cancer.