- AI
- Molecular Imaging
- CT
- X-Ray
- Ultrasound
- MRI
- Facility Management
- Mammography
FDA Clears Catheter with Integrated Intravascular Ultrasound Technology
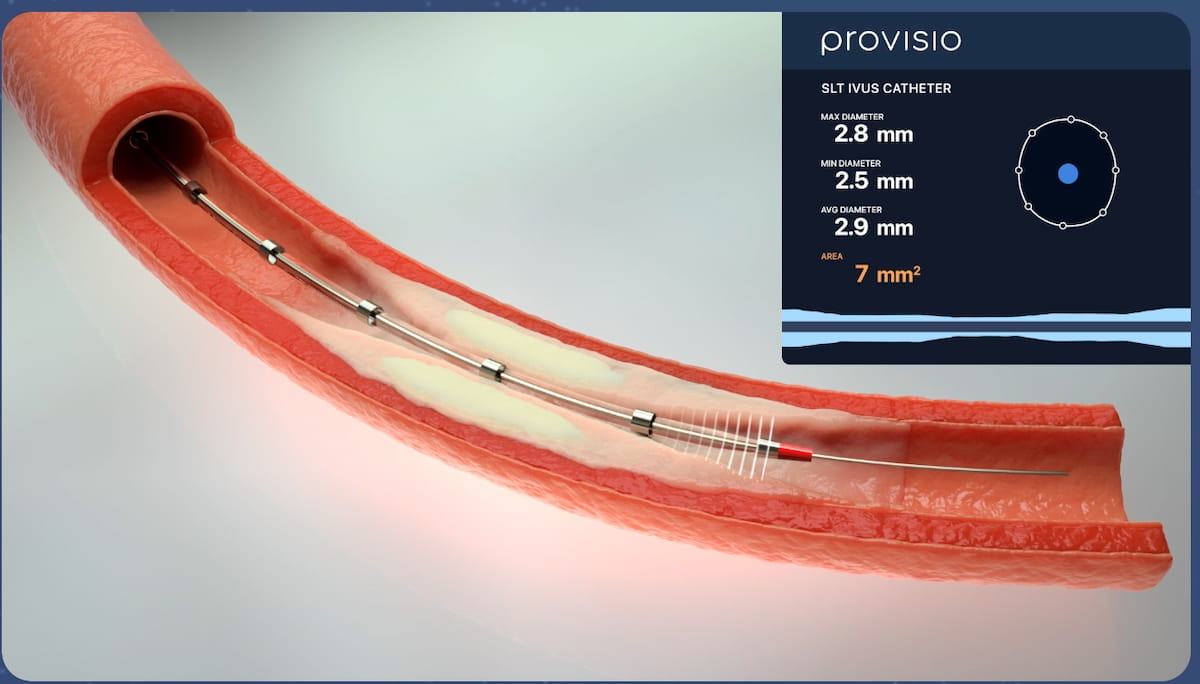
Combining a support crossing catheter with intravascular ultrasound imaging, the Provisio SLT IVUS System offers simultaneous real-time visualization and measurement of vessel lumen.
The Food and Drug Administration (FDA) has granted 510(k) clearance for the Provisio SLT IVUS System, which features a support crossing catheter that integrates intravascular ultrasound, enabling clinicians to assess blood vessel lumen and obtain vessel measurements at the same time through Sonic Lumen Tomography (SLT).
Provisio Medical , the manufacturer of the Proviso SLT IVUS System, said the Provisio SLT IVUS System’s inclusion of the SLT IVUS Support Crossing Catheter provides dual functionality in interventional procedures with an ultrasound transducer array at the distal end of the catheter.
Through the use of Sonic Lumen Tomography (SLT), the newly FDA-cleared Provisio SLT IVUS System provides clinicians with simultaneous real-time visualization and measurement of vessel lumen during interventional procedures for patients with peripheral vascular disease. (Image courtesy of Provisio Medical.)
Incorporating intravascular ultrasound capability into a front-line support crossing catheter may help simplify workflows, reduce iodinated contrast media (ICM) and reduce radiation exposure for patients being treated for peripheral vascular disease (PVD), according to Provisio Medical.
"Clinical outcomes in peripheral vascular disease have consistently been shown to benefit from accurate intravascular measurements, yet adoption has been limited by the additional procedure time and training required to interpret images" noted S. Eric Ryan, M.D., the chief executive officer of Provisio Medical. "Thanks to the ease-of-use of SLT IVUS, which can be incorporated more efficiently in the peripheral vascular workflow, we believe there is the possibility of increased adoption and therefore improved outcomes for many more patients with potentially devastating peripheral vascular disease."
Leading Breast Radiologists Discuss the USPSTF Breast Cancer Screening Recommendations
May 17th 2024In recognition of National Women’s Health Week, Dana Bonaminio, MD, Amy Patel, MD, and Stacy Smith-Foley, MD, shared their thoughts and perspectives on the recently updated breast cancer screening recommendations from the United States Preventive Services Task Force (USPSTF).
Multicenter CT Study Shows Benefits of Emerging Diagnostic Model for Clear Cell Renal Cell Carcinoma
May 15th 2024Combining clinical and CT features, adjunctive use of a classification and regression tree (CART) diagnostic model demonstrated AUCs for detecting clear cell renal cell carcinoma (ccRCC) that were 15 to 22 percent higher than unassisted radiologist assessments.
CT Study: AI Algorithm Comparable to Radiologists in Differentiating Small Renal Masses
May 14th 2024An emerging deep learning algorithm had a lower AUC and sensitivity than urological radiologists for differentiating between small renal masses on computed tomography (CT) scans but had a 21 percent higher sensitivity rate than non-urological radiologists, according to new research.