
Catch up on a variety of new FDA clearances in radiology from the past week.

Catch up on the top radiology content of the past week.

While researchers saw no significant difference in metastasis-free survival with the use of perioperative apalutamide in patients undergoing radical prostatectomy based on conventional imaging alone, the addition of PSMA PET imaging revealed a 20 percent higher likelihood of metastasis-free survival with adjunctive apalutamide.

The MIM Contour ProtegeAI+ 2.0 software reportedly offers new AI-powered contouring models for brain MRIs and pelvic CTs.

In comparison to short-axial field of view PET/CT scanners, a total body PET/CT imaging platform with a long axis field of view detected more lesions in 68 percent of patients while reducing scan times by 83 percent, according to new research presented at the SNMMI conference.

In a recent interview, Phillip Kuo, MD, discussed new comparative multicenter prospective research, presented at the SNMMI conference, examining the urinary radioactivity of two PSMA PET radiotracers 18F-piflufolastat and 18F-flotufolastat.

Prior vaccination for prostate cancer-specific antigens was associated with more than double the rate of two-year overall survival in patients with mCRPC who were treated with 177Lu-PSMA-617, according to new research presented at the SNMMI conference.

Preliminary research findings, presented at the SNMMI conference, suggest that the alpha-emitting radioconjugate ATNM-400 may be a viable alternative for treating PCa that is resistant to androgen receptor pathway inhibitors (ARPIs).

Noting the potential for workload efficiencies in interpreting screening digital breast tomosynthesis (DBT) images, researchers found the use of slab reconstruction technology improved specificity without any impact on sensitivity or cancer detection rate.

Reportedly trained on thousands of cardiac ultrasound images, the Ejection Fraction (EF) AI tool provides automated measurement of left ventricular ejection fraction (LVEF) in seconds.

The AI-powered Elevate Plus reportedly facilitates standardization of routine ultrasound, improves scan time and bolsters image quality.
Emphasizing the capability to evaluate the spatiotemporal distribution of PET radiotracers, the MIM KineticID software may provide greater quantitative insights into oncologic, cardiovascular and neurological disease management.
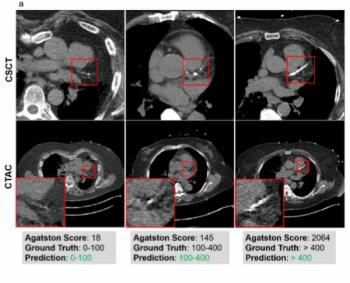
For patients undergoing myocardial perfusion PET/CT, a deep learning model demonstrated greater than 83 percent AUCs for classifying those with low- and high-risk coronary artery calcium (CAC) burden, according to new research presented at the SNMMI conference.

In the latest episode of the Molecular Imaging in Focus series, Isabelle Boileau, PhD, discussed preliminary research, presented at the SNMMI conference, which suggests the utility of a novel tau PET imaging agent that may help detect chronic traumatic encephalopathy (CTE) in living patients.

Do you embrace challenges and change or settle and wallow in your diagnostic comfort zone?
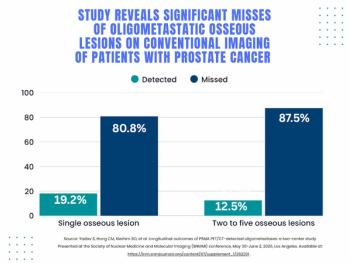
For patients with oligometastatic prostate cancer detected with PSMA PET, researchers found that nearly 78 percent of these patients had negative findings on conventional imaging, according to a study presented at the Society for Nuclear Medicine and Molecular Imaging (SNMMI) conference.

In a recent interview, Shannon Youngblood, EdD, MSRS, CNMT, RT(CT)(ARRT, shared her perspectives on advocacy for nuclear medicine technologists, scope of practice challenges, theranostics and improving access to PET imaging in rural communities.

Catch up on the top radiology content of the past week.

The Cone-Beam CT (CBCT) Perfusion software reportedly provides CT-quality perfusion maps during neurointerventional procedures.

Catch up on the most-well viewed radiology content in May 2026.

In a recent interview, Josh Gurewitz and Ajit Shankaranarayanan, PhD, discussed the utility of AI-powered software, including the recently FDA-cleared SubtleHD(PET), for bolstering efficiency and quality with PET imaging.

Catch up on the most well-viewed video interviews from Diagnostic Imaging in May 2026.

A randomized controlled trial found that combining Disney characters with soothing ambient elements for pediatric MRI reduced anxiety levels by 43 percent in children under 10 years of age.

Emerging CCTA research affirms the importance of going beyond LDL reduction in the evaluation and treatment of patients with chronic coronary syndrome.
The SubtleHD(PET) software reportedly facilitates enhanced image quality, provides improved SUVmax quantitative accuracy and compatibility with all FDA-approved radiotracers.

After the U.S. Preventive Services Task Force (USPSTF) updated its biennial mammography screening recommendations to begin at 40 years of age, women in their 40s accounted for approximately 50 percent of all screening mammography exams, according to new study findings to be presented at the American Society of Clinical Oncology (ASCO) conference.

In recent interviews, Emanuel Kanal, MD, and Tobias Gilk, MRSO, MRSE, discuss new multicenter research examining pediatric MRI safety as well as key challenges and preventive measures in this patient population.

Catch up on the top AI-related news and research in radiology over the past month.
While there are pros and cons to consider, offering a direct pay service for second opinions on imaging may emerge as a viable, middleman-free alternative for radiologists.

Catch up on the top radiology content of the past week.