Adjunctive AI Powered Tool for Tuberculosis Detection Gets FDA's Breakthrough Device Designation
With reported increases in tuberculosis cases after the COVID-19 pandemic, the qSpot-TB adjunctive artificial intelligence (AI) device may facilitate improved diagnosis of the disease on chest X-rays.
The Food and Drug Administration (FDA) has granted breakthrough device designation to qSpot-TB, an artificial intelligence (AI)-powered device, which may enhance tuberlosis (TB) detection on chest X-rays.
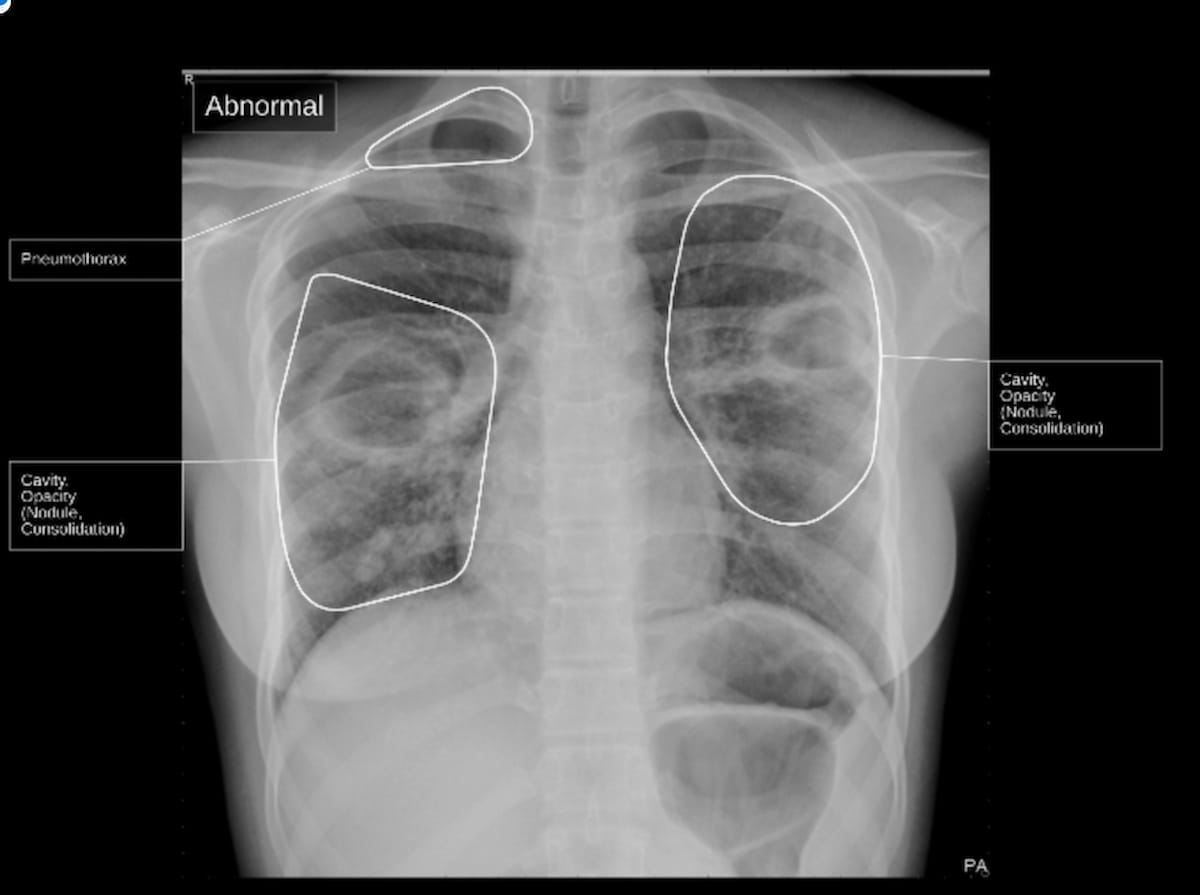
Qure.ai, the developer of qSpot-TB, said the device localizes signs of TB on chest X-rays and provides a conclusion of whether TB is present or not.
The emergence of the qSpot-TB device is particularly timely, according to the company, given statistics from the Centers for Disease Control and Prevention (CDC) showing 8,300 reported TB cases in 2022.
The qSpot-TB device, which recently garnered a breakthrough device designation from the FDA, localizes signs of TB on chest X-rays, and provides a conclusion of whether TB is present or not, according to Qure.ai, the developer of qSpot-TB. (Image courtesy of Qure.ai)
"The increase in TB cases in USA is a reminder about the importance of collective global efforts to continue the fight against the disease until eliminated. We cannot let our guard down. Innovative technology is a crucial component for accelerated progress to successfully end TB globally,” said Professor Kenneth G. Castro, M.D., a co-director of the Emory Tuberculosis Center, and professor of global health, epidemiology, and infectious disease with the Rollins School of Public Health at Emory University.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.
Stroke MRI Study Assesses Impact of Motion Artifacts Upon AI and Radiologist Lesion Detection
July 16th 2025Noting a 7.4 percent incidence of motion artifacts on brain MRI scans for suspected stroke patients, the authors of a new study found that motion artifacts can reduce radiologist and AI accuracy for detecting hemorrhagic lesions.