FDA clearances may foreshadow hot spots at this year’s RSNA meeting
Latest batch of 510(k)s has something for everyoneFDA clearances surged to 25 during October but fell short of the level reached during the same period last year, when the regulatory agency cleared 30 radiological devices in the
Latest batch of 510(k)s has something for everyone
FDA clearances surged to 25 during October but fell short of the level reached during the same period last year, when the regulatory agency cleared 30 radiological devices in the month preceding the 2000 RSNA meeting.
The clearances were well distributed across modalities: CT, radiotherapy, ultrasound, and x-ray had four each, MR received five, image management had three, and nuclear medicine won a single clearance for a PET/CT scanner enhancement.
Several of the new devices are sure to be showcased on the RSNA exhibit floor. At press time, the FDA had not released public statements describing the 510(k) clearances, but summary sheets and other available documents filled in some of the blanks.
Noteworthy among the CT offerings is an advanced lung analysis product from GE Medical Systems. At last year’s RSNA meeting, GE showed early clinical results of a sophisticated volumetric rendering package that works in concert with its multislice LightSpeed CT scanner. The clearance the FDA granted GE on Oct. 26 could be a logical follow-on to that early work and might mark the beginning of an effort by CT vendors to open up new clinical opportunities in lung screening.
Dose optimization will be the other shoe to fall in CT this year. Philips received FDA clearance on Oct. 1 to begin marketing its DoseRight technology, which adjusts dose to match patient habitus. The market has been waiting for this product since its unveiling in Chicago last year. Its transit through the regulatory system in time for commercialization this year means that Philips has a homegrown technological counterweight to the acquisition of Marconi Medical and its Mx8000. To advance its corporate image, the company needs to possess in-house tools to develop advanced CT technologies. Having a commercial product that addresses dose concerns will help, especially since GE has been selling SmartmA technology, albeit only for its HiSpeed X/i products, and Siemens has been publicizing its Care Dose optimization system since the 1999 European Congress of Radiology.
Two ultrasound products cleared the FDA in October, but neither of the developers of these systems will have a booth at the upcoming meeting, although their equipment may be the subject of discussions on the exhibit floor. The Chinese company Shantou Institute of Ultrasonic Instruments received FDA clearance to sell a high-end portable ultrasound scanner called the CTS-485. Performance specifications include 256 gray-scale, cine-loop, RS232 interface and broadband multifrequency probes, which support applications in cardiology, ob/gyn, and abdominal, thyroid, small parts, uterus, and breast imaging.
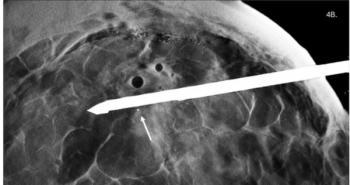
Dynamic Imaging in Scotland received FDA clearance to sell its Diasus. The system, optimized for breast and musculoskeletal imaging, operates over a relatively high and broad frequency range of 5 to 22 MHz. The ultrawide-band linear-array probes produce resolution to as little as 50 microns 2 cm below the skin surface. Such high resolution allows visualization of tears in the tendon, tendon sheaths, and internal fibrillar structure, according to the company. In breast ultrasound, Diasus promises to better define lesion margins and more accurately guide needle biopsy.
UltraGuide will finally be able to promote its MR-Guide 2000 interventional system. Although the system was shown at last year’s RSNA meeting, the Israeli company waited until late September to submit an application to the FDA. A positive response was received within a month, setting the stage for the company to officially launch the product at the upcoming RSNA show.
MR-Guide 2000 could change MR intervention by enabling invasive procedures to be performed inside a conventional closed tube scanner. The guide provides a real-time display and needle locator that helps clinicians maneuver interventional and surgical devices. The display also plots the trajectory of these devices to forecast their path. MR-Guide joins similar products the company markets for use in ultrasound and CT.
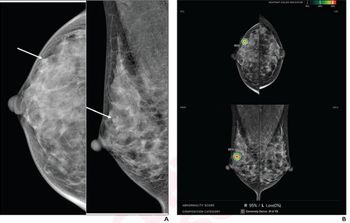
Lorad, a division of Hologic, received FDA clearance on Oct. 24 for its Affinity Mammography System. The screen-film system specifically addresses the market need for a cost-effective product with high-performance characteristics, according to the company. Affinity will be marketed globally and is scheduled to be in full production in the first quarter of 2002.
The FDA disclosed four clearances in early November. Optima URS, a universal radiographic system, promises to boost the U.S. arm of its fledgling Spanish developer Sedecal, which is headquartered in Madrid. Sedecal U.S.A. will offer a wide assortment of x-ray-based systems, from mobile x-ray to radiography and fluoroscopy products. Optima URS, a low-cost universal swivel-arm x-ray system that cleared the FDA on Nov. 2, consists of a floor-to-wall column and a turnable arm that can be positioned at varying heights. Patients can be examined while standing, sitting, or lying down.
Finally, H Innovation received clearance to market its iConnection 3D workstation. This connects seamlessly with the company’s miniPACS/teleradiology system. The newly cleared product supports integrated clinical solutions for online interactive access, processing, review, and distribution of volumetric images.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.