FDA Clears Cardiac AI Software That May Reduce MRI Scan Times by 83 Percent
Powered by deep learning technology, Sonic DL reportedly facilitates the acquisition of cardiac magnetic resonance imaging (MRI) scans at 12 times the speed of conventional MRI systems.
The Food and Drug Administration (FDA) has granted 510(k) clearance to Sonic DL (GE HealthCare), a deep learning software that may reinvent scan time expectations and possibly expand the eligibility of patients for cardiac magnetic resonance imaging (MRI).
GE HealthCare said Sonic DL can improve radiology workflow efficiency and alleviate backlogs with its ability to slash scan times for cardiac MRI by up to 83 percent. Emphasizing the capability of Sonic DL to facilitate real-time functional imaging “as fast as a single heartbeat,” the company noted the artificial intelligence (AI) technology was designed to allow the acquisition of MRI scans at 12 times the rate of conventional MRI.
The accelerated scanning may also allow broader patient access to cardiac MRI, according to Gianluca Pontone, M.D., Ph.D., F.E.S,C., the director of the Perioperative Cardiology Cardiovascular Imaging Department at Centro Cardiologico Monzino in Milano, Italy.
“Sonic DL emerges as a game-changer in the field of cardiac imaging. By capturing images within a single heartbeat, this cutting-edge technology addresses the unique needs of patients who face challenges in breath-holding, suffer from advanced heart failure, or have arrhythmias,” noted Dr. Pontone. “The significance of this capability cannot be overstated as it ensures a smoother and more comfortable experience for patients during MRI exams.”
Here one can see conventional cardiac MRI (left) offering whole heart coverage in one minute and 42 seconds vs. Sonic DL (right), which facilitates whole heart coverage in 59 seconds. Note that rapid imaging on the right reduces motion artifacts for a patient who cannot follow a breath hold command. (Images courtesy of GE HealthCare.)
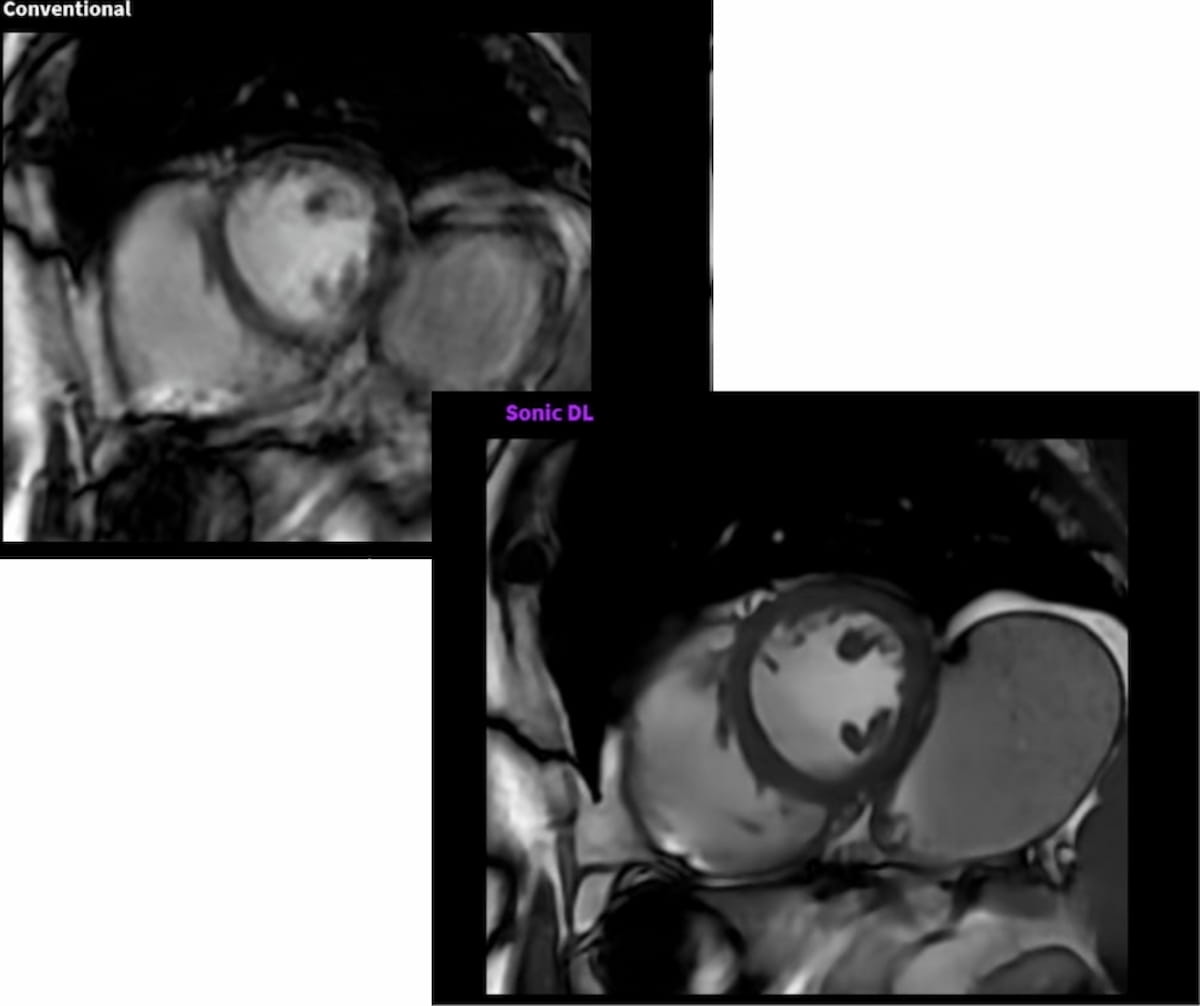
(Editor’s note: For related content, see “FDA Clears AI Software That May Lead to 30-Minute Full-Body MRI Exams,” “Emerging AI Advances in Cardiac Imaging” and “Can Emerging AI-Guided Software Rein in Scan Times for Cardiac MRI?”)
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.
Stroke MRI Study Assesses Impact of Motion Artifacts Upon AI and Radiologist Lesion Detection
July 16th 2025Noting a 7.4 percent incidence of motion artifacts on brain MRI scans for suspected stroke patients, the authors of a new study found that motion artifacts can reduce radiologist and AI accuracy for detecting hemorrhagic lesions.