FDA Clears CT-Based AI Software for Assessing Interstitial Lung Disease
Reportedly trained on thousands of computed tomography scans, the e-Lung software utilizes machine learning to detect and assess the progression of features associated with interstitial lung diseases.
The Food and Drug Administration (FDA) has granted 510(k) clearance for the artificial intelligence-powered e-Lung software, which may enhance assessment for interstitial lung disease (ILD) on computed tomography (CT) scans.1
Incorporating evaluation of vascular structures in the periphery of the lung and reticular opacities, the e-Lung software provides a weighted reticulovascular score (WRVS) that quantifies key contributing factors for ILDs, according to Brainomix, the developer of the e-Lung software.1,2
In recent research, published in the Journal of Respiratory and Critical Care Medicine, researchers found that an e-Lung WRVS threshold of > 15 percent was associated with a greater than fivefold risk of forced vital capacity (FVC) decline. The AI-powered e-Lung software was recentlly granted 510(k) clearance from the FDA. (Image courtesy of Brainomix.)
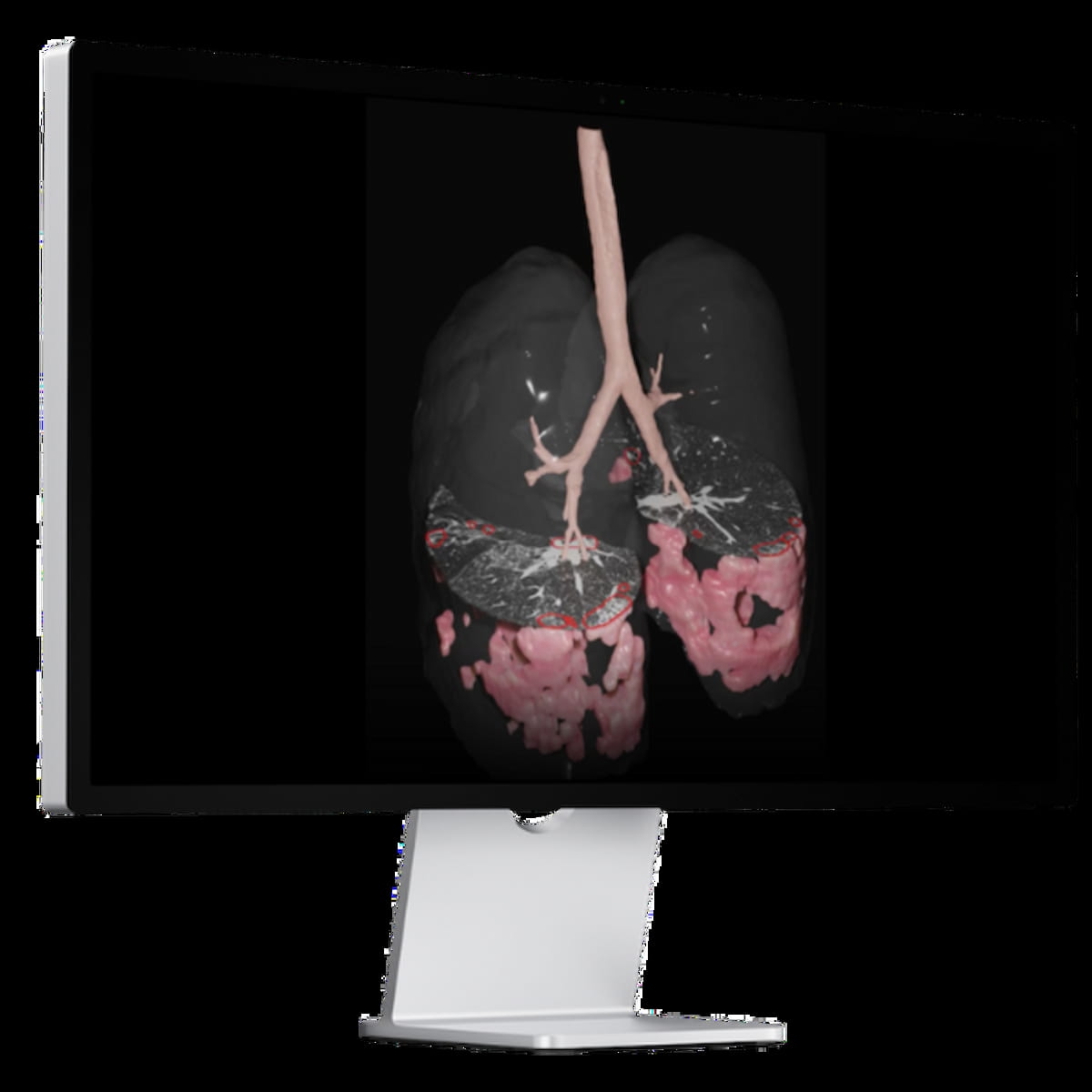
In recent research, published in the Journal of Respiratory and Critical Care Medicine, researchers found that an e-Lung WRVS threshold of > 15 percent was associated with a greater than fivefold risk of forced vital capacity (FVC) decline.2 The study authors also noted that at 52 weeks, WRVS assessment had a 16 percent higher C-index for identifying FVC decline in comparison to conventional FVC assessment (77 percent vs. 61 percent).2
“The preliminary data for e-Lung is impressive and would indicate that we have a promising tool that could help to expedite healthcare delivery and improve clinically meaningful outcomes for patients with lung disease,” noted Deji Adegunsoye, M.D., an assistant professor of medicine and scientific director of the Interstitial Lung Disease Program at the University of Chicago Medicine.
References
1. Brainomix. Brainomix achieves breakthrough with FDA clearance of e-Lung AI software. PR Newswire. Available at: https://www.prnewswire.com/news-releases/brainomix-achieves-breakthrough-with-fda-clearance-of-e-lung-ai-software-302144832.html . Published May 17, 2024. Accessed May 17, 2024.
2. Devaraj A, Ottink F, Rennison-Jones C, et al. E-Lung computed tomography biomarker stratifies patients at risk of idiopathic pulmonary fibrosis progression in a 52-week clinical trial. Am J Respir Crit Care Med. 2024;209(9):1168-1169.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.
Stroke MRI Study Assesses Impact of Motion Artifacts Upon AI and Radiologist Lesion Detection
July 16th 2025Noting a 7.4 percent incidence of motion artifacts on brain MRI scans for suspected stroke patients, the authors of a new study found that motion artifacts can reduce radiologist and AI accuracy for detecting hemorrhagic lesions.