Lunit Partners with Philips on Chest X-ray AI Solution
Lunit’s INSIGHT CXR software will be available on Philips’ X-ray solutions.
Medical artificial intelligence (AI) start-up Lunit announced, during the 2021 European Congress of Radiology (ECR) annual meeting, that its AI software will now be available on Philips Healthcare’s diagnostic X-ray solutions.
For additional ECR 2021 coverage, click here.
This move marks the latest extension of Philips’ AI portfolio in precision diagnosis. It is designed to improve patient outcomes and experience, according to a company statement.
Lunit INSIGHT CXR detects findings and provides abnormality score on a chest X-ray image
Credit: Lunit
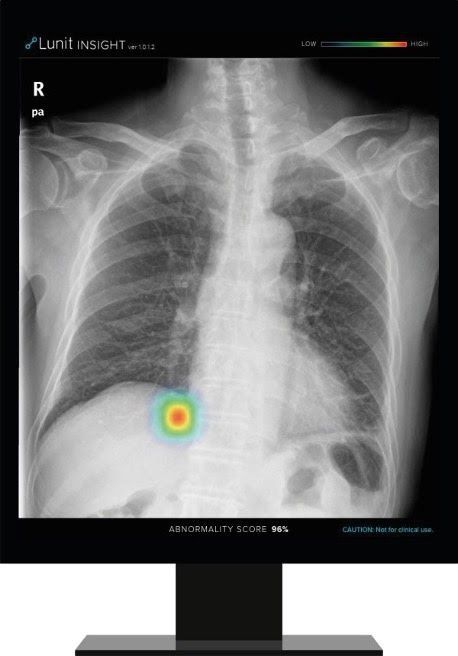
Lunit INSIGHT CXR chest detection suite can identify 10 of the most common findings on chest X-rays by mapping finding locations and displaying a scored calculation of where those findings actually are. By using abnormality scores, the software prioritizes scans, allowing for faster triage of the most critical images.
Based on existing publications, including studies in Radiology and JAMA Network Open, the algorithm can perform with 97 percent to 99 percent accuracy.
This collaboration will potentially accelerate the diagnostic capabilities of facilities, said Daan van Manen, general manager for diagnostic X-ray at Philips.
“Radiology departments and their technologists are continually under pressure. They face high patient volumes, and every improvement in workflow can make a big impact,” Manen said. “Our partnership with Lunit to incorporate their diagnostic AI into our X-ray platform combines with a host of smart workflow features in the Philips Eleva user interface, our common platform across our digital radiography systems that enables a smooth and efficient, patient-focused workflow.”
Lunit INSIGHT CXR is already CE marked and clinically available in Europe, Middle East, Latin America, South East Asia, Australia, and New Zealand. The company expects U.S. Food & Drug Administration clearance this year.
For more coverage based on industry expert insights and research, subscribe to the Diagnostic Imaging e-newsletter here.
Considering Breast- and Lesion-Level Assessments with Mammography AI: What New Research Reveals
June 27th 2025While there was a decline of AUC for mammography AI software from breast-level assessments to lesion-level evaluation, the authors of a new study, involving 1,200 women, found that AI offered over a seven percent higher AUC for lesion-level interpretation in comparison to unassisted expert readers.
Can CT-Based Deep Learning Bolster Prognostic Assessments of Ground-Glass Nodules?
June 19th 2025Emerging research shows that a multiple time-series deep learning model assessment of CT images provides 20 percent higher sensitivity than a delta radiomic model and 56 percent higher sensitivity than a clinical model for prognostic evaluation of ground-glass nodules.
FDA Clears Ultrasound AI Detection for Pleural Effusion and Consolidation
June 18th 2025The 14th FDA-cleared AI software embedded in the Exo Iris ultrasound device reportedly enables automated detection of key pulmonary findings that may facilitate detection of pneumonia and tuberculosis in seconds.