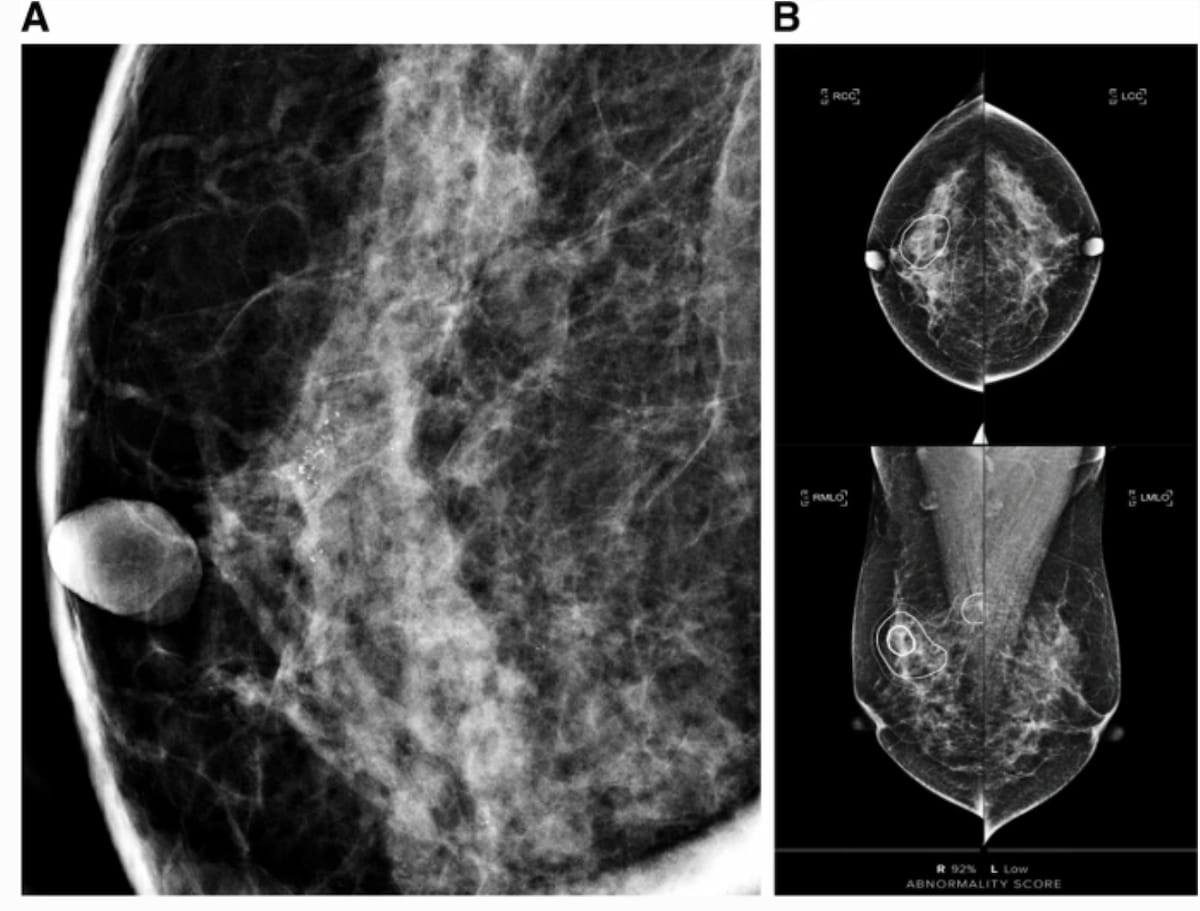
Artificial intelligence (AI) may bolster risk stratification of women with ductal carcinoma in situ as new research demonstrates a link between AI scoring of mammography abnormalities and elevated risks for progression to invasive breast cancer.
For the study, recently published in Insights into Imaging, researchers assessed the ability of an AI-based computer-aided detection/diagnosis (CAD) modality (Lunit Insight for Mammography, Lunit) to predict progression to invasive cancer in 420 women with a total of 440 cases of ductal carcinoma in situ (DCIS) diagnosed with percutaneous biopsy. They noted that 341 of the DCIS cases were detected via mammography. According to the study, 117 cases of DCIS were upgraded to invasive cancer.
For patients with DCIS that was diagnosed with percutaneous biopsy, the researchers found that an AI-CAD score of >50 for mammography abnormalities nearly doubled the likelihood of invasive cancer (1.96 odds ratio (OR)) and an AI-CAD score of >75 more than doubled the likelihood of DCIS progressing to invasive breast cancer (2.306 OR).
“Based on our results, DCIS with higher AI-CAD scores, especially in the highest quartile of >75%, can be considered to have invasive components,” wrote study co-author Jung Hyun Yoon, M.D., who is affiliated with the Department of Radiology at the Severance Hospital and the Research Institute of Radiological Science at the Yonsei University College of Medicine in Seoul, South Korea, and colleagues.
While the researchers found that AI-CAD scores >50 and >75 were not independently prognostic for invasive cancer progression in those with mammography-detected DCIS, they suggested that the high prevalence of calcifications in this cohort (78.6 percent) and the varied presentations of these lesions may have factored into these findings.
“Radiologists encounter difficulties assessing abnormalities with ambiguous, overlapping imaging features, which seems to be the same for AI-CAD as dichotomized or graded AI-CAD scores could not independently predict DCIS upgrade in mammographically detected ones,” noted Yoon and colleagues.
Three Key Takeaways
- AI-CAD scores as predictors. The study suggests that AI-based computer-aided detection/diagnosis (CAD) scores from mammography abnormalities can serve as valuable predictors for the progression of ductal carcinoma in situ (DCIS) to invasive breast cancer. Specifically, AI-CAD scores exceeding 50 and 75 correlate with significantly increased likelihoods of invasive cancer progression, highlighting the potential of AI in risk stratification.
- Combination of features. Combining AI-CAD scores with clinicopathologic features enhances the accuracy of prognosis assessment for women with DCIS. A combination of these factors, particularly an AI-CAD score exceeding 75, more than doubles the likelihood of invasive cancer, according to the study.
- Clinical implications. The findings underscore the significance of AI tools in improving prognosis assessment and guiding treatment decisions for DCIS. Early identification of DCIS with invasive components using AI may aid in selecting appropriate patients for active monitoring or surgical intervention, potentially reducing unnecessary procedures and improving patient outcomes. However, the study acknowledges the need for further research to validate these findings across diverse populations and settings.
However, the study authors pointed out that a combination of clinicopathologic features and an AI-CAD score >75 had more than double the likelihood of invasive cancer in this cohort (2.145 OR). They also noted area under the receiver operating characteristic curves (AUROCs) for total DCIS (69.9 to 70.3 percent) and mammographically detected DCIS (67.7 to 68.8 percent) for the AI-CAD model (which was designed for breast cancer detection) that were comparable to AUROCs for AI algorithms specifically trained for invasive upgrade prediction for those with DCIS.
“In this context, comparable outcomes can be considered highly promising, as this achievement suggests that pre-existing AI technologies can be used to predict invasive upgrade,” added Yoon and colleagues.
The study authors emphasized that AI tools that can enhance prognosis assessment for women with DCIS could play a key role with patient management.
“Predicting DCIS upgrade before surgery is crucial for surgical decision-making, as the overall incidence of axillary lymph node metastasis is around 5% but rises to 20% for preoperatively underestimated lesions. This highlights the need for performing sentinel biopsies on patients with a strong preoperative upgrade prediction to prevent additional completion surgery,” maintained Yoon and colleagues. “Moreover, given the current trend (toward) less aggressive treatment for low-risk DCIS including the omission of surgery, accurately identifying DCIS with invasive components becomes vital in selecting the right patients for active monitoring.
(Editor’s note: For related content, see “Mammography Study: AI Improves Breast Cancer Detection and Reduces Reading Time with DBT,” “Mammography Study: Deep Learning Emphasis on Bilateral Dissimilarity May Clarify AI Assessments of Breast Cancer Risk” and “Can AI Automate BPE Assessment of Dense Breasts on MRI?”)
Beyond the inherent limitations of a single-center retrospective study, the authors noted the entire cohort was comprised of Asian women and over 88 percent had dense breasts. They conceded that one breast radiologist determined the mammographic features of DCIS as well as final BI-RADS assessments.