MR elastography inspires new wave of hepatic imaging
Just in time for a looming onset of new liver disease, MR elastography has arrived to employ principles as old as palpation and as new as cross-sectional imaging to create an accurate, noninvasive way to diagnose and stage hepatic fibrosis and other liver disorders.
Just in time for a looming onset of new liver disease, MR elastography has arrived to employ principles as old as palpation and as new as cross-sectional imaging to create an accurate, noninvasive way to diagnose and stage hepatic fibrosis and other liver disorders.
Chronic liver disease and cirrhosis are major public health problems worldwide. In 2004, these conditions were associated with nearly 40,000 deaths and a cost of at least $1.4 billion for medical services in the U.S. alone. Furthermore, these figures are expected to increase based on an aging population, the growing epidemic of obesity, and the continuing trend toward end-stage liver disease among individuals with chronic hepatitis C.1
The major biological process responsible for clinical liver disease is progressive hepatic fibrosis. Liver biopsy has been considered the gold standard for detecting hepatic fibrosis. However, the use of liver biopsy in large populations will not be practical. Specifically, there are limitations with the technique, including high cost, poor acceptance by patients, and measurement errors, that will prevent its more widespread use.2 To date, the lack of a suitable noninvasive alternative to liver biopsy has represented a major obstacle for managing the many diseases that cause hepatic fibrogenesis.
Conventional imaging technologies, such as CT, MRI, and ultrasound, are useful for diagnosing the complications of advanced liver disease (varices, ascites, spenomegaly), but they do not allow reliable identification of hepatic fibrosis unless it is very advanced. The addition of Doppler flow measurements to ultrasound, texture analysis to CT, or diffusion-weighted imaging or dynamic contrast enhancement to MRI have been reported to improve the diagnosis of moderately advanced hepatic fibrosis. But these techniques have not been shown to be sensitive in the detection of early- and midstage disease.1
Physicians have traditionally used palpation as a valuable clinical diagnostic technique to detect many disease processes-including cirrhosis-in which the liver typically becomes very hard to the touch. Recent laboratory research has shown that the stiffness of liver tissue is strongly correlated with the extent of fibrosis.3,4
Recognizing the potential value of this type of information in clinical medicine, researchers have worked to develop methods to quantitatively image the stiffness of tissue. One of the most promising of these technologies is magnetic resonance elastography.5,6 Originally developed for applications such as cancer imaging and biomechanics,7,8 MRE is emerging as a potent modality for diagnosing hepatic fibrosis in patients with chronic liver disease.9-11
MRE is not just a promising concept; it is now applied clinically every day at the Mayo Clinic. This technology has been successfully translated from our research laboratories where it was developed and was made available to enhance patient care at the Mayo Clinic in mid-2007. Since that time, MRE has been included in the abdominal MR examinations of more than 500 patients to assess various aspects of liver disease. Ruling out hepatic fibrosis with MRE has become one of the most frequent requests in referrals for abdominal MRI. With a negative predictive value of 97% for excluding hepatic fibrosis, MR elastography is especially valuable in triaging patients for liver biopsy.
WORKING PRINCIPLES
MRE uses low-frequency mechanical waves to probe the elastic properties of tissue. The technology requires modest software and hardware upgrades to a conventional MR scanner.
The procedure follows three steps. First, mechanical waves (from 40 Hz to 200 Hz) are generated in the body using an MRI-compatible acoustic driver device. For liver MRE, this is typically done by placing a simple passive drumlike device over the anterior abdominal wall. This "passive driver" is connected by a plastic tube to a speakerlike "active driver," which is located outside the magnet room (Figure 1).
In the second step, these waves are imaged using a special phase-contrast MR imaging sequence. This sequence provides remarkable sensitivity, visualizing wave motions as small as 1/1000th of a millimeter, by synchronizing the motion-encoding gradients with the applied vibration.
The typical acquisition time for hepatic MRE is approximately 15 seconds, well within a breath-hold. In these images, the pattern of wave propagation is governed by local tissue stiffness. Therefore, the third and final step of this technology is to process the wave data using a specialized mathematical "inversion" algorithm to generate an "elastogram," a diagnostic image that quantitatively depicts the stiffness of tissue in the cross section interrogated (Figure 2).
12
The average liver stiffness value can be calculated by assessing regions of interest throughout the image. Normal liver tissue has a stiffness of approximately 2 kiloPascals (kPa), which is very soft compared with most other tissues in the body and similar to that of subcutaneous fat. Because the acquisition time is very short, the addition of MRE for liver evaluation into a conventional MRI examination protocol adds very little to the typical total examination time of 30 to 45 minutes.
DIAGNOSING HEPATIC FIBROSIS
Multiple studies have shown that MR elastography can be used to accurately diagnose hepatic fibrosis in patients with liver disease,9-11, 13 In one study, for instance, MR elastography was performed in 35 healthy individuals and 48 patients with varying degrees of biopsy-proven chronic liver disease (Figure 3).11 The mean liver stiffness value for normal individuals was 2.2 kPa. For the entire group of patients with liver disease, the mean liver stiffness value was 5.8 kPa. The mean liver stiffness measurements increased with greater degrees of liver fibrosis.
In the meantime, noninvasive evaluation of hepatic fibrosis has emerged as the first mainstream clinical
Statistical analysis of the data indicated that if 2.93 kPa is used as the upper limit of normal for liver tissue stiffness, then MRE is estimated to have a sensitivity of 98% and specificity of 99% for differentiating any stage of liver fibrosis from normal liver tissue. If liver stiffness is over 4.89 kPa, then the sensitivity and specificity for diagnosing patients with moderate to severe fibrosis are estimated to be 86% and 85%, respectively.
Fatty liver is a common occurrence in patients with chronic liver disease. Some observers have speculated that the accumulation of fat in liver tissue might make it softer, thereby negating the increased stiffness that would otherwise indicate the presence of fibrosis. However, research has now shown that the presence or absence of hepatic steatosis has little or no influence on liver tissue stiffness and therefore does not affect the MRE-based assessment of hepatic fibrosis.11
ULTRASOUND ELASTOGRAPHY
An ultrasound-based technology for quantitatively assessing hepatic stiffness has also been introduced in the last several years in Europe and other parts of the world. Known as transient ultrasound elastography, this technique employs a specialized device that uses 1D ultrasound imaging to measure the propagation speed of a mechanical wave generated by a vibrator applied to the body wall.14
Multiple studies have shown that this technology has excellent sensitivity (>85%) and specificity (>90%) for detecting cirrhosis in patients with chronic liver disease.14 Meta-analysis of the existing literature, however, indicates that diagnostic performance of transient ultrasound elastography is lower in patients with early-stage hepatic fibrosis, increased fatty infiltration of the liver on biopsy, or high body mass index (≥28 kg/m2). Transient ultrasound elastography generally cannot be performed in patients with ascites, which is common in chronic liver disease. In this respect, MRE appears to have an advantage, in that experience has shown no limitations in obtaining diagnostic elastograms in patients with marked obesity or ascites.15,16
DIAGNOSING FOCAL HEPATIC LESIONS
Inspired by the successful application of MRE to detect hepatic fibrosis noninvasively, investigators have sought to evaluate its utility for characterizing focal hepatic lesions. Noninvasive characterization of focal liver lesions is typically based on findings in unenhanced T1- and T2-weighted images and dynamic enhancement patterns after contrast administration. Since MRE can easily be included in any abdominal MRI protocol-before or after contrast-enhanced imaging-immediate elastographic analysis of lesions incidentally identified at MRI can be done.
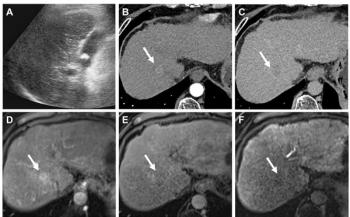
In a recent preliminary study, MRE evaluated 29 patients with 44 liver tumors (14 metastatic lesions,
12
hepatocellular carcinomas, nine hemangiomas, five cholangiocarcinomas, three cases of focal nodular hyperplasia, and one hepatic adenoma) (Figure 4).
17
Malignant liver tumors had significantly higher mean shear stiffness values (10.1 kPa) than benign tumors (2.7 kPa) and normal liver tissue. Given the large difference in mean shear stiffness between benign tissues and malignant lesions, this preliminary study suggests that MRE promises to be useful as a tissue characterization method that might complement traditional MRI criteria in differentiating between benign and malignant lesions. A threshold shear stiffness value of greater than 5 kPa may prove to be a useful criterion in characterizing malignant lesions (Figure 5).
IMAGING PORTAL HYPERTENSION
Portal hypertension is the hemodynamic consequence of cirrhosis and accounts for the clinical complications seen in advanced disease, including esophageal variceal bleeding. While recent studies have demonstrated a potential relationship between liver stiffness as measured by ultrasound-based transient elastography and portal pressure, the association between development of esophageal varices and specific liver stiffness threshold values remains inconsistent.
In contrast, preliminary data using MRE have identified spleen stiffness as a potential imaging marker for portal hypertension.18 Both spleen and liver stiffness appeared highly correlated in a study comparing 12 healthy individuals with 38 patients affected by varying stages of liver disease.
It was also observed that esophageal varices are more likely to develop in patients with mean spleen stiffness values above 10.5 kPa. Further studies to verify these observations in conjunction with portal pressure measurements are ongoing.
TECHNICAL CONSIDERATIONS
Experience has shown that conventional MR scanners can readily be upgraded with the hardware and software necessary to perform MR elastography. The cyclic motion-encoding gradients necessary to image propagating shear waves can be incorporated into almost any type of MRI pulse sequence, including gradient echo, spin echo, and echo planar methods.
MRE sequences are compatible with throughput-enhancing techniques such as parallel imaging and with motion artifact reduction techniques such as spatial presaturation, gradient moment nulling, and respiratory gating. MRE has been implemented on low-, mid-, and high-field MR scanners.
A key requirement for MR elastography is a suitable MRI-compatible acoustic driver system. Ideally, the device for generating waves in the body should be compact, capable of being flexibly oriented against the body, and entirely nonmetallic so that it does not generate artifacts.
There are a variety of options for locating the driver system against the body wall for evaluating the liver. However, several investigators have shown that applying vibrations to the inferior chest wall over the liver will generate shear waves that propagate mainly transversely in a large portion of the liver. This allows a dramatic reduction in imaging time because valid elastograms can be generated from individual axial wave images, which can be acquired in 15 seconds or less.
The alternative to this approach is to fully image the 3D pattern of wave propagation within the liver, but this can require as much as 20 minutes of acquisition time.13 Investigators are using acceleration techniques such as echo planar and parallel imaging to reduce the time required for full 3D MRE, and this advance will be particularly important for imaging other organs in the abdomen and focal lesions, where it may not be possible to generate a simplified wave field.
In the meantime, noninvasive evaluation of hepatic fibrosis has emerged as the first mainstream clinical
The wave amplitudes generated within the body for MRE are very low, typically less than 0.1 mm in displacement. Recent studies have shown that the amplitude of vibration that is being used for MRE is well below the safety limits established by the European Union for chronic occupational vibration exposure.19
EXPANDING CLINICAL ROLE
Investigators have barely scratched the surface in exploring the potential applications of MR elastography (Figure 6). In liver imaging alone, there are numerous opportunities to explore with this new imaging modality. Other potential applications range from oncology to neurologic disease to assessing biomaterials. MR elastography offers a new range of imaging biomarkers that will take years to fully explore.
application of this new imaging technology. In this role, MRE offers a more comfortable, safer, less expensive, and potentially more accurate alternative to liver biopsy.
References
1. Talwalkar JA, Yin M, Fidler JL, et al. Magnetic resonance imaging of hepatic fibrosis: emerging clinical applications. Hepatology 2008;47(1):332-342.
2. Bedossa P, Dargère D, Paradis V. Sampling variability of liver fibrosis in chronic hepatitis C. Hepatology 2003;38(6):1449-1457.
3. Yeh WC, Li PC, Jeng YM, et al. Elastic modulus measurements of human liver and correlation with pathology. Ultrasound Med Biol 2002;28(4):467-474.
4. Georges PC, Hui JJ, Gombos Z, et al. Increased stiffness of the rat liver precedes matrix deposition: implications for fibrosis. Am J Physiol Gastrointest Liver Physiol 2007;293(6):G1147-1154.
5. Muthupillai R, Lomas DJ, Rossman PJ, et al. Magnetic resonance elastography by direct visualization of propagating acoustic strain waves. Science 1995;269(5232): 1854-1857.
6. Muthupillai R, Ehman RL. Magnetic resonance elastography. Nat Med 1996;2(5):601-603.
7. McKnight AL, Kugel JL, Rossman PJ, et al. MR elastography of breast cancer: preliminary results. AJR 2002;178(6):1411-1417.
8. Ringleb SI, Bensamoun SF, Chen Q, et al. Applications of magnetic resonance elastography to healthy and pathologic skeletal muscle. J Magn Reson Imaging 2007;25(2): 301-309.
9. Rouviere O, Yin M, Dresner MA, et al. MR elastography of the liver: preliminary results. Radiology 2006; 240(2):440-448.
10. Huwart L, Peeters F, Sinkus R, et al. Liver fibrosis: non-invasive assessment with MR elastography. NMR Biomed 2006;19(2):173-179.
11. Yin M, Talwalkar JA, Glaser KJ, et al. Assessment of hepatic fibrosis with magnetic resonance elastography. Clin Gastroenterol Hepatol 2007;5(10):1207-1213.
12. Manduca A, Oliphant TE, Dresner MA, et al. Magnetic resonance elastography: non-invasive mapping of tissue elasticity. Med Image Anal 2001;5(4):237-254.
13. Huwart L, Sempoux C, Salameh N, et al. Liver fibrosis: noninvasive assessment with MR elastography versus aspartate aminotransferase-to-platelet ratio index. Radiology 2007;245(2):458-466.
14. Talwalkar JA, Kurtz DM, Schoenleber SJ, et al. Ultrasound-based transient elastography for the detection of hepatic fibrosis: systematic review and meta-analysis. Clin Gastroenterol Hepatol 2007;5(10):1214-1220.
15. Talwalkar JA. Elastography for detecting hepatic fibrosis: options and considerations. Gastroenterology 2008 Jun 11. [Epub ahead of print] 16. Huwart L, Sempoux C, Vicaut E, et al. Magnetic resonance elastography for the non-invasive staging of liver fibrosis. Gastroenterology: in press.
17. Venkatesh SK, Yin M, Glockner JF, et al. MR elastography of liver tumors: preliminary results. AJR 2008;190(6):1534-1540.
18. Talwalkar JA, Yin M, Venkatesh S, et al. Spleen stiffness measurement by magnetic resonance elastography is an independent predictor of esophageal varices in patients with compensated cirrhosis. Hepatology 2007;46(4 Suppl S):575A.
19. Ehman EC, Rossman PJ, Kruse SA, et al. Vibration safety limits for magnetic resonance elastography. Phys Med Biol 2008;53(4):925-935.
Dr. Talwalkar is an associate professor of medicine at the Center for Advanced Imaging Research at the Mayo Clinic in Rochester, MN. Dr. Yin is a senior research fellow at the same institution.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.