Vessel imagers focus on plaque characterization
Plaque rupture is the main cause of acute coronary syndromes, which are associated with high mortality and morbidity. The prevalence of plaque rupture in acute coronary syndrome is more than 70%.1 Rupture-prone plaques are characterized by a large core of extracellular lipid with a thin fibrous cap (Figure 1). Inflammation, particularly in the plaque shoulder, causes the fibrous cap to thin and results in disruption of the atherosclerotic plaque.
Plaque rupture is the main cause of acute coronary syndromes, which are associated with high mortality and morbidity. The prevalence of plaque rupture in acute coronary syndrome is more than 70%.1 Rupture-prone plaques are characterized by a large core of extracellular lipid with a thin fibrous cap (Figure 1). Inflammation, particularly in the plaque shoulder, causes the fibrous cap to thin and results in disruption of the atherosclerotic plaque. When the fibrous cap ruptures, thrombogenic substances are exposed to the blood. The developing thrombus, which consists of platelet aggregates, leukocytes, fibrin, and red blood cells, obstructs the vessel, causing myocardial infarction or stroke.
Plaque rupture is seen as a morphologic lumen abnormality on many different imaging modalities. Intravascular ultrasound,2 angioscopy,3 and optical coherence tomography (OCT)4 are all popular tools for detecting plaque rupture. Early detection of vulnerable plaque, including rupture-prone plaque, is necessary to identify vulnerable patients. Both IVUS and angioscopy have detected multiple plaque rupture in patients with acute coronary syndrome.2,3 The development of virtual histology IVUS and integrated backscatter IVUS has improved plaque characterization significantly,5,6 as has the advent of techniques such as thermography, palpography, and elastography.
Multislice CT and MRI have been shown to visualize the coronary vasculature with high accuracy compared with invasive angiography.7,8 MSCT is also able to perform plaque characterization.7,9,10 The spatial resolution of MSCT was previously poor compared with other invasive plaque imaging devices, but the latest generation of equipment, including dual-source CT, has changed this. The spatial resolution of DSCT can reach 0.3 to 0.4 mm.11
Both noninvasive and invasive modalities can consequently be used to detect plaque rupture and to monitor the healing process of ruptured plaques.
INVASIVE VIEW OF RUPTURE
Coronary angiography, which is essentially luminography, can give only limited information on vessel wall pathology. A retrospective analysis of angiography showed that two-thirds of patients with acute myocardial infarction had a previous stenosis ( < 50% occlusion) at the site of the culprit lesion before plaque rupture.12 When the plaque is ruptured without occlusion, coronary angiography depicts the culprit lesion as ulceration. Angiography cannot identify these sites as vulnerable, however, or monitor the healing process.
Gray-scale IVUS can detect major features of ruptured plaque, such as ulceration, cavity, intimal flap, thrombus, and aneurysm (Figure 2).13 It can also partially detect the fibrous cap.13 Plaques identified on IVUS as having a larger echolucent area or thinner fibrous plaque appear to be prone to rupture.14
Spectral analysis of the radiofrequency component of backscatter IVUS, or virtual histology IVUS, has been developed for real-time in vivo plaque classification.5 This technique classifies fibrofatty plaque, fibrous plaque, necrotic core, and dense core from histologic data (Figure 3A). Which component contributes most to the plaque's vulnerability remains controversial. Thin-cap fibroatheroma lesions are the most prevalent substrate of plaque rupture. Research has shown that the percentage volume of atheroma and the mean percentage of necrotic core in stable patients and in patients with acute coronary syndrome are no different.15
Integrated backscatter IVUS is another new technique based on RF data analysis that is able to evaluate a lipid core accurately and quantitatively (Figure 3C).5,6 Integrated backscatter values for plaque components have been described as follows:16
- thrombus: > -88 and -80
- intimal hyperplasia or lipid core: > -73 and lesser than or equal to -63
- fibrous tissue: > -63 and lesser than or equal to -55
- mixed lesions: > -55 and lesser than or equal to -30
- calcification: > -30 and lesser than or equal to -23
When using either virtual histology or integrated backscatter IVUS, the lumen border should be assessed on gray-scale images (3B and 3D).
Lesions causing acute coronary syndrome are typically seen on angioscopy as yellow plaque with an irregular surface covered by a thrombus.3,10 The yellow color represents the lipid-rich core and thin fibrous cap, and the intensity of yellow color reflects vulnerability (Figure 4).10 Asakura et al found the prevalence of yellow plaques and thrombus in lesions causing acute coronary syndrome to be 90% and 81%, respectively.3 Patients with acute coronary syndrome had widespread disease in all three major coronary arteries and multiple yellow intact plaques.3
OCT is a high-resolution imaging method with 10-nm spatial resolution.4 There is good correlation between OCT and histologic measurements of the fibrous cap.17 OCT can identify mural and luminal morphologies, including lumen dimensions, plaques, thrombi, dissections, and tissue flaps.4 It can also detect ruptured plaque.18 The limited penetration of OCT (1 to 2 mm) might make it difficult to image an entire vulnerable plaque using this technique.
NONINVASIVE DETECTION
MSCT can detect coronary stenosis with high accuracy when compared with invasive coronary angiography.7 One of the main strengths of MSCT is its extremely high negative predictive value.7 A major weakness, however, is the difficulty of interpreting plaque characteristics and distribution from gray-scale CT. CT number (defined as water = 0 and air = -1000) can be used for tissue characterization, but region of interest or profile curve was not comprehensive for the analysis. To counter this, we developed a new method of plaque color mapping (Plaque Map) that is based on CT number (Figure 5).9,10
MSCT is regarded as a valuable tool for detecting acute coronary syndrome in the emergency room.10 Limitations include the use of contrast media and exposure to ionizing radiation.
The advent of DSCT may provide a more precise noninvasive way of examining coronary plaque.11 Current DSCT systems are equipped with two x-ray tubes mounted at 90 degrees to one another. Beta blockers are not required to control heart rate, given the improved temporal resolution (83 msec). Spatial resolution of DSCT is 0.3 to 0.4 mm, while that of 64-slice MSCT is 0.5 mm at best. DSCT can demonstrate ruptured plaque with lipid pool.19 The benefits of a dual-energy setup for detecting plaque should be the subject of future study.
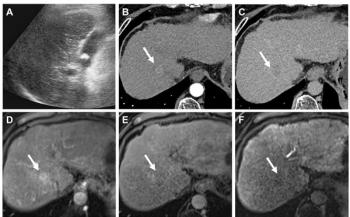
Noninvasive detection of coronary stenosis by MR coronary angiography is also possible. A patient-based analysis performed by Sakuma et al found the sensitivity, specificity, positive predictive values, and negative predictive values compared with x-ray coronary angiography to be 82%, 90%, 88%, and 86%, respectively.8 Diagnostic accuracy in the individual segments were 78%, 96%, 69%, and 98%, respectively.8 MR coronary angiography can detect morphologic abnormalities of silent plaque rupture, such as double lumen and positive remodeling (Figure 6).20 MRA can potentially depict atherothrombotic plaque components, including lipid, fibrous tissue, calcium, and thrombus. Further improvements to temporal and spatial resolution are needed, though, before MRA can be used to detect coronary artery plaque.
VULNERABLE PLAQUE FOLLOW-UP
Sudden coronary occlusion leading to acute coronary syndrome is often preceded by a period of plaque instability and thrombus formation. This may start days or weeks before symptomatic onset.21 We have devised a strategy for detecting coronary artery plaque ruptures in high-risk patients using DSCT. The culprit lesion itself should be treated invasively. Ruptured plaque at nonculprit lesions should be treated with a plaque stabilization therapy such as statin drugs, clopidogrel, or aspirin. Follow-up should be performed on DSCT (Figure 7).22
Rioufol et al treated 14 patients with statin and antiplatelet agents for 28 lesions of spontaneous coronary plaque rupture without significant stenosis detected by IVUS. They found that half of the lesions were healed or the grade of stenosis was diminished for two-year follow-up. Spontaneous coronary plaque rupture without significant stenosis, detected on first acute coronary syndrome diagnosis, healed without significant plaque modification in 50% of cases with medication.23
A two-year follow-up period was used for this study.
The combination of noninvasive and invasive imaging devices may provide the optimal strategy for studying the pathogenesis of acute coronary syndrome and applying the best therapy to patients.
Assisting in the preparation of this manuscript were Dr. Christoph D. Garlichs, Dr. Dieter Ropers, Dr. Alexander Kuhlmann, Dr. Ulrike Ropers, and Prof. Dr. Werner G. Daniel from the department of cardiology, University of Erlangen-Nuernberg; Dr. Tilman Rau, Dr. David Lukas Wachter, and Dr. Valentina Campean from the department of pathology, University of Erlangen-Nuernberg; Dr. Atsushi Hirayama and Dr. Kazuhisa Kodama from the cardiovascular division, Osaka Police Hospital; and Prof. Masatsugu Hori from the department of cardiovascular medicine, Osaka University Graduate School of Medicine, Suita.
REFERENCES
- Naghavi M, Libby P, Falk E, et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: Part I. Circulation 2003;108(14):1664-1672.
- Hong MK, Mintz GS, Lee CW, et al. The site of plaque rupture in native coronary arteries; a three-vessel intravascular ultrasound analysis. J Am College Cardiol 2005;46(2):261-265.
- Asakura M, Ueda Y, Yamaguchi O, et al. Extensive development of vulnerable plaques as a pan-coronary process in patients with myocardial infarction: an angioscopic study. J Am College Cardiol 2001;37(5):1284-1288.
- Gerckens U, Buellesfeld L, McNamara E, Grube E. Optical coherence tomography (OCT). Potential of a new high-resolution intracoronary imaging technique. Herz 2003;28(6):496-500.
- Nair A, Kuban BD, Tuzcu EM, et al. Coronary plaque classification with intravascular ultrasound radiofrequency data analysis. Circulation 2002;106(17):2200-2206.
- Komiyama N, Berry GJ, Kolz ML, et al. Tissue characterization of atherosclerotic plaques by intravascular ultrasound radiofrequency signal analysis: an in vitro study of human coronary arteries. Am Heart J 2000;140(4):565-574.
- Achenbach S. Computed tomography coronary angiography. J Am College Cardiol 2006;48(10):1919-1928.
- Sakuma H, Ichikawa Y, Chino S, et al. Detection of coronary artery stenosis with whole-heart coronary magnetic resonance angiography. J Am College Cardiol 2006;48(10):1946-1950.
- Komatsu S, Hirayama A, Omori Y, et al. Detection of coronary plaque by computed tomography with the novel plaque analysis system, 'Plaque Map,' and comparison with intravascular ultrasound and angioscopy. Circ J 2005;69(1):72-77.
- Komatsu S, Ueda Y, Omori Y, et al. Diagnosis of vulnerable plaque and vulnerable patient by coronary angioscopy and multi-detector row computed tomography-from invasive to non-invasive plaque imaging. Vascular Disease Prevention 2006;3(4):319-325.
- Achenbach S, Ropers D, Kuettner A, et al. Contrast-enhanced coronary artery visualization by dual-source computed tomography-initial experience. Europ J Radiol 2006;57(3):331-335.
- Ambrose JA, Winters SL, Stern A, et al. Angiographic morphology and the pathogenesis of unstable angina pectoris. J Am College Cardiol 1985;5(3):609-616.
- Jensen LO, Mintz GS, Carlier SG, et al. Intravascular ultrasound assessment of fibrous cap remnants after coronary plaque rupture. Am Heart J 2006;152(2):327-332.
- Ge J, Chirillo F, Schwedtmann J, et al. Screening of ruptured plaques in patients with coronary artery disease by intravascular ultrasound. Heart 1999;81(6):621-627.
- Rodriguez-Granillo GA, Garcia-Garcia HM, McFadden EP, et al. In vivo intravascular ultrasound-derived thin-cap fibroatheroma detection using ultrasound radiofrequency data analysis. J Am College Cardiol 2005;46(11):2038-2042.
- Kawasaki M, Takatsu H, Noda T, et al. In vivo quantitative tissue characterization of human coronary arterial plaques by use of integrated backscatter intravascular ultrasound and comparison with angioscopic findings. Circulation 2002;105(21):2487-2492.
- Kume T, Akasaka T, Kawamoto T, et al. Measurement of the thickness of the fibrous cap by optical coherence tomography. Am Heart J 2006;152(4):755.e1-4.
- MacNeill BD, Jang IK, Bouma BE, et al. Focal and multi-focal plaque macrophage distributions in patients with acute and stable presentations of coronary artery disease. J Am College Cardiol 2004;44(5):972-979.
- Komatsu S, Daniel WG, Achenbach S. Demonstration of clinically silent plaque rupture by dual-source computed tomography. Eur Heart J 2007; February 6 [E-pub ahead of print].
- Komatsu S, Murakawa T, Shinohe T, et al. Detection of silently ruptured plaque by coronary magnetic resonance. Int J Cardiol 2006;113(3):430-431.
- Rittersma SZ, van der Wal AC, Koch KT, et al. Plaque instability frequently occurs days or weeks before occlusive coronary thrombosis: a pathological thrombectomy study in primary percutaneous coonary intervention. Circulation 2005;111(9):1160-1165.
- Komatsu S, Hirayama A, Ueda Y, et al. Coronary ruptured plaque mimicking spontaneous coronary dissection in a young woman. Int J Cardiol 2006;113(2):288-289.
- Rioufol G, Gilard M, Finet G, et al. Evolution of spontaneous atherosclerotic plaque rupture with medical therapy: long-term follow-up with intravascular ultrasound. Circulation 2004;110(18):2875-2880.
Dr. Komatsu is a clinical fellow in the cardiology department at University of Erlangen-Nuernberg in Erlangen, Germany, and a senior consultant in the cardiovascular division at Osaka Police Hospital, Japan. Prof. Dr. Amann is a senior consultant in the pathology department, and Prof. Dr. Achenbach is a senior consultant in the cardiology department, both at the University of Erlangen-Nuernberg.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.