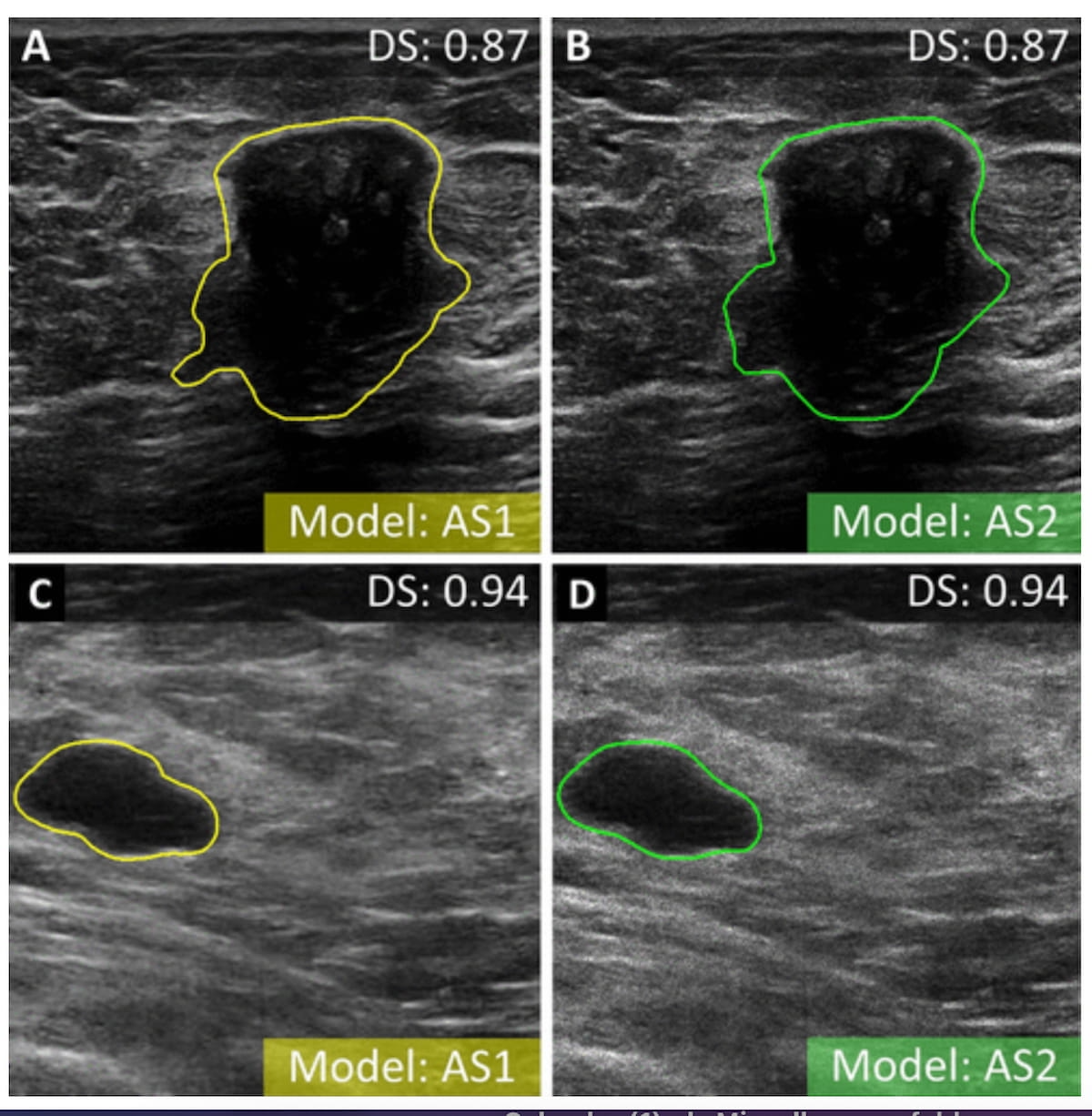
Emerging research suggests the combination of radiomic and autoencoder features may bolster ultrasound sensitivity and specificity for diagnosing breast cancer in a real-time setting.
For the retrospective study, recently published in Radiology, researchers employed ultrasound data from 1,191 women (mean age of 61) to develop breast cancer ultrasound classification models that incorporated radiomics, autoencoder-based features or mixed features.1 The study authors noted the image-based features were derived from: local segmentation by radiologists; nnU-net local segmentation; whole image; automated bounding box assignment from radiologist segmentation; and automated bounding box assignment from nnU-Net.
The researchers found that the most effective model was a mixed model derived with bounding boxes from local segmentation provided by radiologists. Trained with 23 mixed features, the model had a 90 percent area under the receiver operating characteristic curve (AUC), 81 percent sensitivity and 87 percent specificity, according to the study authors. The study findings also revealed an 88 percent positive predictive value (PPV) and a 78 percent negative predictive value (NPV) for the radiologist bounding box model.1
“We showed that bounding box–based categorization can better discriminate between malignant and benign breast lesions, independently of the used image-based features,” wrote lead study author Zuzanna Anna Magnuska, Ph.D., who is affiliated with the Institute for Experimental Molecular Imaging at RWTH Aachen University in Aachen, Germany, and colleagues.
The study authors noted that the tumor bounding box model offered 14 percent higher sensitivity rate and 10 percent higher specificity rates than estimates from previous research examining the use of breast ultrasound without computer-assisted diagnosis.1-3
Additionally, in external validation testing, the researchers noted no statistical difference between AUCs for the aforementioned model, a radiologist with 20 years of experience and a radiologist with three years of experience.
“We believe that our framework could become reliable assistance for standard (ultrasound)-based breast examination performed by any users. It also might have an impact on reducing intraobserver variability,” added Magnuska and colleagues.
Three Key Takeaways
1.sImproved Sensitivity and specificity: The study demonstrated that a mixed model using automated bounding box assignments generated by local tumor segmentation from radiologists and autoencoder features achieved 81 percent sensitivity and 87 percent specificity for diagnosing breast cancer via ultrasound.
2. Bounding box-based categorization: The researchers noted the use of tumor bounding boxes that include peritumoral tissue enhanced the model's ability to distinguish between malignant and benign breast lesions.
3. Potential for reducing intraobserver variability. The proposed framework could assist in standard ultrasound-based breast examinations, potentially reducing intraobserver variability, allowing radiologists of varying experience levels to achieve similar diagnostic results.
(Editor’s note: For related content, see “Breast Ultrasound Study: AI Radiomics Model May Help Predict Lymphovascular Invasion in Breast Cancer Cases,” “Mammography Study Shows Supplemental Ultrasound Has Higher Sensitivity than Adjunctive AI in Dense Breasts” and “Predicting DCIS Upgrade to Invasive Breast Cancer: Can Contrast-Enhanced Ultrasound Have an Impact?”)
In an accompanying editorial, Manisha Bahl, M.D., M.P.H., FSBI, noted that the four FDA-cleared AI algorithms for breast ultrasound were trained on static two-dimensional images.4 In addition to real-time application with the model highlighted in the study. Dr. Bahl said the use of tumor bounding boxes provides insight beyond models that emphasize lesion segmentation.
“Unlike detailed lesion segmentation, lesion bounding boxes include adjacent breast tissue and this provide important information about the peritumoral environment, which can aid in the discrimination between benign and malignant lesions,” noted Dr. Bahl, an associate professor at Harvard Medical School and a fellow of the Society of Breast Imaging.
Beyond the inherent limitations of a retrospective study, the authors acknowledged that the reported specificity rates with their model may have been influenced by assessing a single cross-section of ultrasound images that included the most suspicious features of breast lesions. While noting the inclusion of six different data sets in the study, the researchers conceded the ground truth for the study was obtained by single radiologists for each of the data sets.
References
1. Magnuska ZA, Roy R, Palmowski M, et al. Combining radiomics and autoencoders to distinguish benign and malignant breast tumors on US images. Radiology. 2024 Sep; 312(3):e232554. doi: 10:1148/radiol.232554.
2. Geisel J, Raghu M, Hooley R. The role of ultrasound in breast cancer screening: the case for and against ultrasound. Semin Ultrasound CT MR. 2018;39(1):25-34.
3. Aristokli N, Polycarpou I, Themistocleous SC, ophocleous D, Mamais I. Comparison of the diagnostic performance of magnetic resonance imaging (MRI), ultrasound and mammography for detection of breast cancer based on tumor type, breast density and patient’s history: a review. Radiography. 2022;28(3):848-856.
4. Bahl M. Combining AI and radiomics to improve the accuracy of breast US. Radiology. 2024 Sep;312(3):e241795. doi: 10.1148/radiol.241795.