FDA Clears AI-Powered Prostate Volume Measurement for Handheld Ultrasound
Offering expedited calculation of prostate volume, Clarius Prostate AI is reportedly the first AI-enabled prostate measurement tool to garner FDA clearance for use with handheld ultrasound.
The Food and Drug Administration (FDA) has granted 510(k) clearance for Clarius Prostate AI, an artificial intelligence (AI) application for handheld ultrasound that enables rapid calculation of prostate volume.
In contrast to manual processes with traditional ultrasound, clinicians can utilize the Clarius app to access Clarius Prostate AI, which highlights the prostate gland and facilitates caliper placement to measure prostate volume, according to Clarius, the developer of Clarius Prostate AI.
The newly FDA-cleared Clarius Prostate AI, which enables rapid calculation of prostate volume with handheld ultrasound, is currently available with Clarius handheld ultrasound scanners that allow customization for urology exams. (Image courtesy of Clarius Mobile Health.)
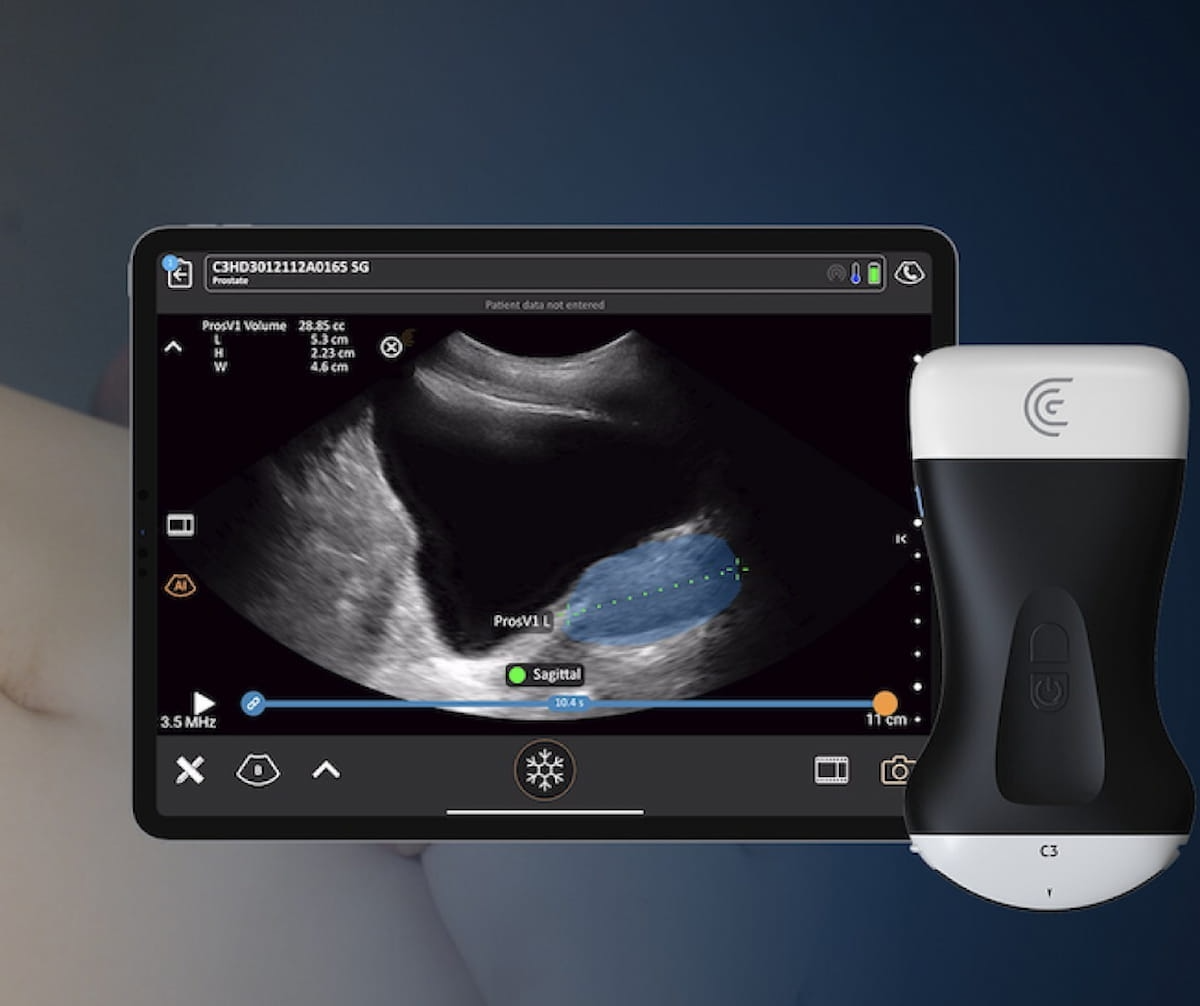
The company said Clarius Prostate AI, which is being showcased at the American Urological Association (AUA) conference April 26-29, also calculates prostate-specific antigen (PSA) density by combining the prostate volume measurement with the inputted patient’s PSA level.
"95% of clinicians we recently surveyed believe AI-powered measurements would save them time and allow them to see more patients. Manual ultrasound prostate volume measurements are repetitive and time-consuming, regardless of experience. With Clarius Prostate AI, what used to take several minutes now takes seconds, no matter your experience level,” noted Sarah Leverett, the vice president of marketing at Clarius Mobile Health.
Clarius noted that Clarius Prostate AI is currently available with Clarius handheld ultrasound scanners, including the C3 HD3 for transabdominal ultrasound and the EC7 HD3 for transrectal imaging, that offer customization for urology exams.
FDA Clears Virtually Helium-Free 1.5T MRI System from Siemens Healthineers
June 26th 2025Offering a cost- and resource-saving DryCool magnet technology, the Magnetom Flow.Ace MRI system reportedly requires 0.7 liters of liquid helium for cooling over the lifetime of the device in contrast to over 1,000 liters commonly utilized with conventional MRI platforms.