FDA clearances skyrocket in May to highest level in seven months
X-ray, CT, and MRI include substantial new devicesThe number of devices cleared for marketing in the U.S. boomed in May to the highest level since last October. A total of 31 devices passed review at the regulatory agency, a sharp
X-ray, CT, and MRI include substantial new devices
The number of devices cleared for marketing in the U.S. boomed in May to the highest level since last October. A total of 31 devices passed review at the regulatory agency, a sharp rise from the 21 that were cleared in April and the 24 in March.
Nine of the most recent 31 were related to MR, though all but one was a coil. CT and x-ray devices accounted for five clearances each. Ultrasound, radiotherapy, and image management each had four.
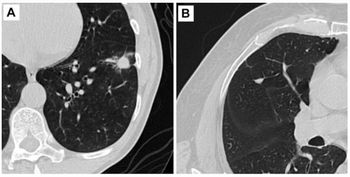
One system straddles two categories: CT and x-ray. The C-arm, called 3D Accu-I-tomo, is designed to acquire a 360 degrees rotational sequence of the head and neck. Developed by J. Morita Manufacturing of Kyoto, Japan, it can operate in either CT or fluoroscopy mode. When functioning as a CT, the device acquires a 3D matrix of the target volume, then displays both 2D and 3D views of the volume. When in fluoroscopy mode, the device provides dynamic 2D images.
The company, founded 86 years ago, specializes in dental products, but the device cleared by the FDA goes beyond dentistry. In its FDA application, the company listed dentists as well as physicians and x-ray technologists among the primary users. The fluoroscopy mode, for example, is suited to surgical applications involving the ear, nose, and throat, as well as the temporomandibular joint, according to Morita.
The C-arm assembly is mounted on a support column, which has an x-ray control panel and emergency stop switch. Remote control is provided to assist in patient positioning. The C-arm assembly includes an image intensifier and x-ray tube. The control box, including operational lights and an x-ray emission button, is designed for separate placement from the rest of the system. A Windows-based PC controls the device.
GE Medical Systems' colonography option is more in the CT mainstream. The image analysis software uses spiral DICOM images to generate endoluminal views that can be interactively accessed by the operator. The purpose is to screen the colon for polyps, masses, cancers, and other lesions. The package renders 2D and 3D images, bookmarks suspected lesions, and allows synchronized viewing of supine and prone data sets.
Toshiba America Medical Systems received clearance to market its Aquilion Super 4 CT, an upgrade of its quadslice Aquilion system. The major improvements are an enhanced computer subsystem and an improved patient couch. Still pending FDA clearance, according to the company, is an upgraded gantry that will allow 400-msec rotation. Toshiba plans to market the integrated system, after clearance, as the Aquilion Super 4 FX (fast exam).
GE was cleared in May to start marketing its Propeller Imaging Option for MRI. The option is a fast spin-echo (FSE)-based pulse sequence. The sequence provides improved signal to noise and contrast to noise compared with traditional FSE with comparable resolution and scan time, according to the company. The Propeller Imaging Option may be used to differentiate tissues with restricted diffusion from those with normal diffusion, a distinction now routinely achieved using diffusion-weighted echo-planar imaging (EPI).
The new technology is designed for use in anatomical regions where significant susceptibility differences exist between adjacent structures, such as tissue-air and tissue-bone interfaces. Such regions in the brain include the cerebellum and internal auditory canal. Vertebrae are also an example, and other high-susceptibility regions include tissue near metallic implants and artificial joints.
Propeller Imaging can augment the standard EPI-based diffusion imaging by providing improved image quality in areas of high susceptibility. This may help the trained physician to visualize pathology in areas traditionally obscured by susceptibility or motion artifacts, according to GE.
Two of the May x-ray clearances stand out. The Philips Integris Allura Flat Detector release 1.2 system is an upgrade to the first Philips angiographic system exhibited at the RSNA meeting last December (SCAN Special Report 12/25/03). Like its predecessor, release 1.2 is intended for use in diagnostic and interventional procedures, as well as pacemaker implantations and electrophysiology.
Canon's new CXDI-50G digital detector is designed for use in portable radiography systems. The 35 x 43-cm amorphous silicon panel captures data for initial display on a preview monitor. The data are then transmitted through a DICOM-compatible digital network for printing.
Two PACS were cleared in May. The iPACS Prism from RealTimeImage is designed to be Internet-friendly for easy deployment over local and wide area networks. The system is a module that will be offered under different brand names, depending on customer implementation.
The Kodak DirectView PACS/Image Management System is a scalable image/data management solution for hospitals and related institutions, including diagnostic workstations on local and wide area networks as well as enterprise data storage centers. The DirectView PACS also provides services that will allow remote sites access to diagnostic images and patient data using industry standard interfaces, such as Web browsers. The system was designed using an open architecture that allows proprietary and off-the-shelf software to be integrated with off-the-shelf hardware configured to meet the needs of individual customers.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.