GE HealthCare Unveils Head-Only MRI Device at ISMRM Conference
For neuroradiologists, the Signa Magnus 3T MRI system may offer enhanced visualization of the brain microstructure, microvasculature, and function.
Offering a combination of advanced gradient technology, deep-learning algorithms and short echo times, GE HealthCare is showcasing the new Signa Magnus 3T MRI at the International Society for Magnetic Resonance in Medicine (ISMRM) Annual Meeting in Singapore, Asia.
Featuring a gradient strength of 300 mT/m and slew rate of 750 T/m/s, the Signa Magnus is dedicated to head-only MRI imaging and reportedly provides enhanced imaging detail of the brain’s function, microstructure, and microvasculature, according to GE HealthCare.
Reportedly offering enhanced imaging detail of the brain’s function, microstructure, and microvasculature, the head-only 3T MRI system Signa Magnus provides higher b-value diffusion with short echo times, according to GE HealthCare, the manufacturer of the device. (Photo courtesy of GE HealthCare.)
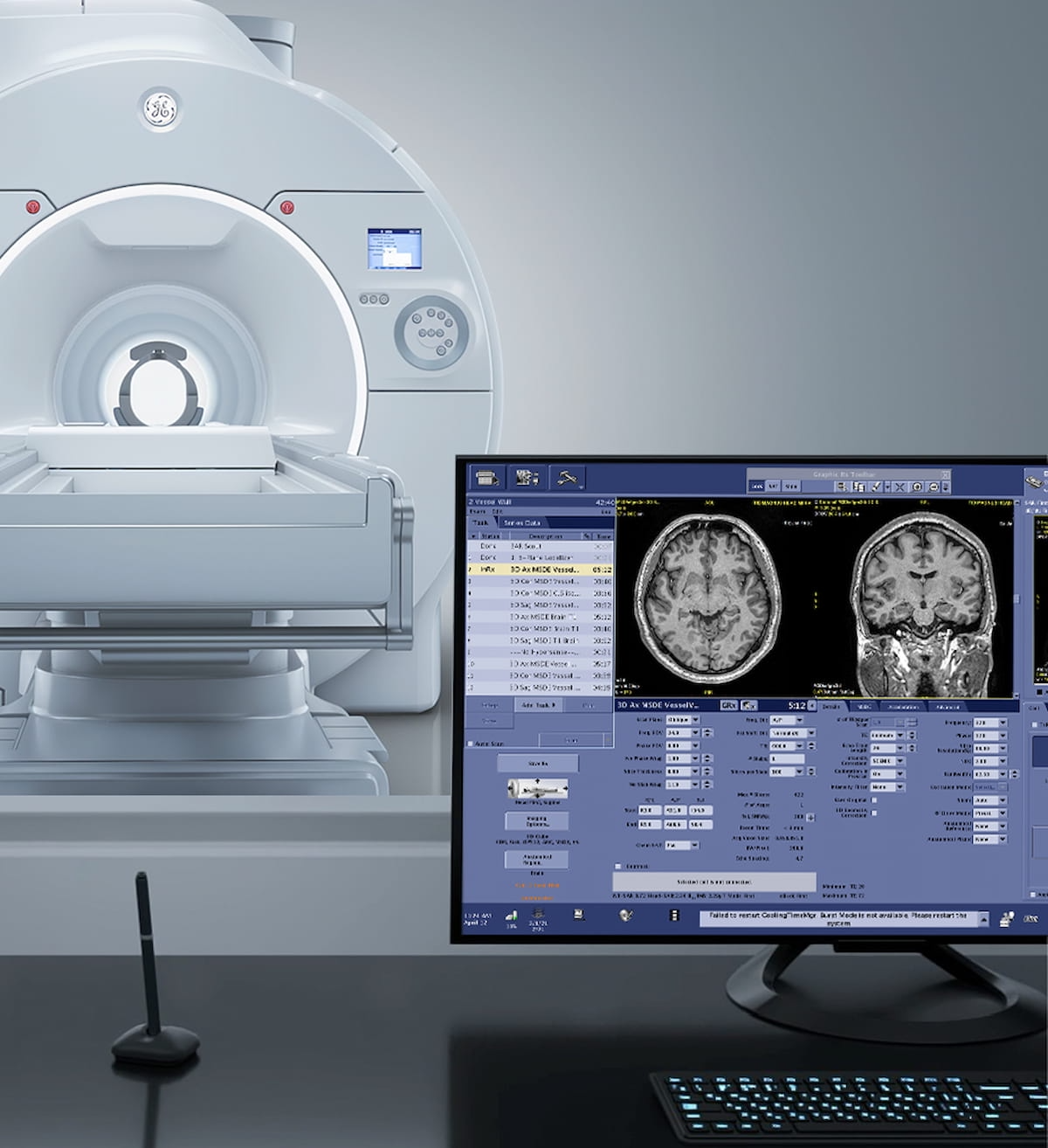
In addition to deep-learning algorithms to bolster advanced imaging techniques, the company said another key attribute of the Signa Magnus MRI system is the capability to achieve higher b-value diffusion with short echo times.
"With Signa Magnus, we are not just exploring the possibility of providing the tool; we are setting new benchmarks in medical research and future clinical patient care.” said Kelly Londy, the chief executive officer of MR for GE HealthCare. “This innovation underscores our commitment to R&D and our collaborations with academia, pushing the boundaries of what's possible in MR imaging. The potential impact of Signa Magnus on patient outcomes and our understanding of the human brain is profound."
GE HealthCare noted that the Signa Magnus system is pending 510(k) clearance from the Food and Drug Administration (FDA).
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.
Key MRI Findings Predictive of Treatment Response for Unresectable Hepatocellular Carcinoma
July 14th 2025For patients with unresectable hepatocellular carcinoma, a pre-treatment MRI finding of LI-RADS tumor in vein was associated with over an 86 percent lower likelihood of responding to transcatheter arterial chemoembolization (TACE) and targeted immunotherapy.