Diagnostic Imaging Europe
- Diagnostic Imaging Europe Vol 26 No 6
- Volume 26
- Issue 6
MR-guided ultrasound ablation gains ground
The therapeutic potential of focused ultrasound was first appreciated almost 70 years ago.
The therapeutic potential of focused ultrasound was first appreciated almost 70 years ago. It was not until the early 1990s, however, that the modality was coupled with MRI to perform the first MR-guided focused ultrasound procedure.1,2
MR-guided focused ultrasound is now accepted as an option for treating for uterine fibroids, and it is being evaluated for the treatment of breast, liver, prostate, and brain tumors. The safety and efficacy of MRgFUS for pain management in cases of skeletal metastases and for the treatment of facet joint syndrome are also being examined in clinical trials. Noninvasive targeted drug delivery, in which focused ultrasound activates drugs at sites of pathology, shows particular promise for the future.
High-intensity focused ultrasound delivers destructive thermal energy to targeted tissues within the body. Thermal energy deposited at the focus of the ultrasound beam denatures proteins and causes coagulative necrosis, leading to cell death. The optimal target temperature for treatment of soft tissue lesions is 140º F (60º C).
The ultrasound beam is focused tightly to treat volumes between 2 × 2 × 8 mm3 and 10 × 10 × 45 mm3. Multiple sonications are needed to treat an entire lesion. Sonications are performed at frequencies of 1 to 1.5 MHz. The transducer frequency, power, target area, and cooling time between sonications can be varied by the operator to ensure that the ablation is consistent across the target, irrespective of tissue type or vascularity.3 Only tissues at the focus of the ultrasound beam are ablated, while areas outside the path of the beam can be avoided.
Focused ultrasound procedures can be guided by either diagnostic ultrasound or MRI. MRgFUS allows the inherently superb diagnostic capabilities of MRI to be exploited and lesions that are visible only on MRI can be identified and targeted.3 MRI can also produce near-real-time thermal maps showing target tissue heating, whereas procedures performed under diagnostic ultrasound guidance cannot be monitored so closely. Near-real-time feedback on tissue heating allows MRgFUS treatments to be tailored to the individual patient.4 Operators can also use software to define “no-go areas” in which sonications are blocked, such as the spinal cord, nerves, bowel, and cutaneous scars, to avoid inadvertent ablation and damage to healthy tissue.
PROCEDURAL POINTERS
The procedure is usually performed under conscious sedation, but general anesthesia may be used if suspended respiration is required. The part of the body containing the target lesion is positioned in a bath containing degassed water and the focused ultrasound probe (Figure 1). A planning scan is performed to pinpoint the lesion’s position, and these geometric coordinates are then coupled with the probe’s positioning system. The focused ultrasound beam, the size and location of which are controlled by the operator, passes through the water bath and into the patient. A test sonication is performed to check that everything is positioned correctly and the equipment is functioning as it should before starting full treatment with multiple sonications.
Near-real-time temperature changes at the target lesion and surrounding tissues, with subsecond temporal resolution, are monitored using phase-shift thermal mapping on a color scale. The temperature-sensitive MRI data are obtained during gradient echo image acquisition. T2-weighted images and pre- and postcontrast T1-weighted images are obtained at the end of the planned series of sonications. Ablated areas are assumed to correlate with unperfused tissue seen on postcontrast images.
Although MRgFUS has enormous potential for treating many soft tissue and skeletal pathologies, it may not be appropriate if a suitable acoustic window is not available. It may not be safe to sonicate the target if the spinal cord and large nerves, overlying ribs, lung tissue, parts of the bowel, and/or cutaneous scars lie within the path of the ultrasound beam. Patients with lesions inside organs that are subject to respiratory movement may require general anesthesia and periods of suspended respiration during sonication.
TARGET LIST
Many soft tissue lesions respond to MRgFUS.
Uterine fibroids. Most MRgFUS procedures to date have been for the treatment of uterine fibroids. Small fibroids may be treated successfully with no additional hormonal therapy. Patients with larger fibroids, ones more than 10 cm in diameter, will typically receive a gonadotropin-releasing hormone agonist for several months prior to MRgFUS (
MRgFUS is a highly effective, noninvasive, long-term treatment for uterine fibroids. Its costs are similar to those of uterine artery embolization and surgical myomectomy but the risks and side effects are considerably reduced.
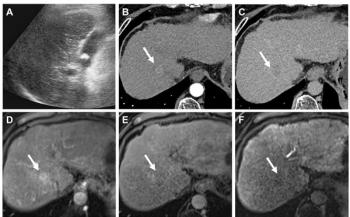
Liver lesions. Primary and secondary liver malignancies are a common clinical presentation. Surgical treatment may, however, be unsuitable or carry excessive risks, while chemotherapy is associated with a multitude of unpleasant and toxic side effects. Many ablative treatments for liver tumors are available, including radiofrequency (RF) ablation, laser ablation, and cryotherapy, and these interventions are frequently extremely effective. The downside is that they require large-bore needles to be inserted into the liver lesion, resulting in potential complications such as bleeding and tumor seeding in the needle tract.
Liver lesions can also be ablated effectively with MRgFUS. This method is totally noninvasive, a benefit that is particularly relevant in patients with poor liver function and defective coagulation. Patients have reported little or no significant postprocedural pain following MRgFUS treatment of liver lesions.3 Initial data from clinical trials are promising and show that the efficacy of MRgFUS is comparable to that of RF and laser ablation. Full results are awaited.
The main difficulty with using MRgFUS to ablate liver lesions is that the target may be difficult to access. The presence of overlying ribs has proved challenging, particularly for lesions in the right lobe under the hemidiaphragm. Liver treatments currently require short periods of suspended respiration at the time of sonication. The development of software that can compensate for respiratory movement will hopefully eliminate the need for general anesthesia and suspended respiration during treatment of liver lesions. The development of small focused ultrasound probes that can fit between the ribs will also allow a greater proportion of liver lesions to be treated with MRgFUS.
Renal lesions. Minimally invasive, curative treatment of small renal tumors that produces little damage to adjacent normal renal tissue is of particular value as an effective alternative to surgery. Initial studies of focused ultrasound ablation of renal tumors have shown mixed results.5 Although MRgFUS of renal lesions is not performed at the moment, this development is likely following successful MRgFUS treatment of other solid organ malignancies.
Breast tumors. Breast cancer is the most common new cancer diagnosed in women in Europe (there were 370,100 new cases in 2004), and it is also the most common cause of cancer death in Europe. Diagnoses are frequently made when the disease is at an early stage, thanks to the success of breast screening programs. The standard treatment for breast cancer typically involves surgery followed by local radiotherapy. The disease is much less likely to recur if patients receive a combination of lumpectomy and local radiotherapy, rather than surgery alone.6
The first MRgFUS treatments of breast lesions were performed in 2001. Treatment achieved partial tumor destruction of 25% to 50%. Subsequent results have improved considerably, with reported tumor destruction reaching 97% ± 3% (Figure 3).7 Any residual viable tumor tends to be found at the lesion edge, hence recommendations that a 5-mm margin around the lesion be included in the treatment field.6 Successful, pain-free treatments have been performed without the use of any analgesia, although some operators use local anesthesia at the treatment site. Trials into MRgFUS plus adjuvant radiotherapy as an alternative to lumpectomy with radiotherapy are also under way.
MRgFUS treatment is generally considered unsuitable for lesions close to the skin, nipple, or chest wall; in cases where there is more than one focal lesion per breast quadrant; for lesions with an extensive intraductal component on needle biopsy; for patients with lymphovascular invasion; or for those who have had previous local treatment.6 MRgFUS may be of value in patients with benign lesions, such as fibroadenomata, who would like the palpable lump to be removed but do not want to be left with a scar. This benefit of a scarfree outcome also applies to patients with malignant breast lesions.
Transcranial MRgFUS. Traditional cranial surgery is associated with a certain amount of collateral damage when gaining access. Any treatment that does not require a craniotomy immediately reduces patient morbidity. Accurate targeting and treatment of an intracranial lesion is of paramount importance. MRI can achieve this through high-resolution imaging of both the lesion and adjacent structures.
Intracranial ultrasound is hampered by the absorption of acoustic energy in the bone matrix, as well as reflection at the inner and outer cortices of the cranium. The level of acoustic energy transmitted to the target lesion within the brain is reduced substantially.8 A specially designed ultrasound probe with a large surface area, together with sonication frequencies of 0.6 to 0.7 MHz, helps prevent overheating in the cranium while ensuring that the lesion is not underheated.8
Clinical trials of transcranial MRgFUS in neoplastic brain lesions are still at an early stage. Initial results suggest, however, that it could potentially be used in combination with some standard forms of therapy, or even replace them altogether.9
In a trial where transcranial MRgFUS was used for noninvasive medial thalamotomy in the treatment of intractable, therapy-resistant neuropathic pain (Figure 4), patients’ pain was reduced by 68% (mean average) two days after the procedure.9 Three-month follow-up results are awaited to see whether the pain relief was permanent.
Focused ultrasound also causes a temporary breakdown in the blood-brain barrier, an effect that has been exploited to deliver drugs into the brain.10,11 This effect is more reliable and reproducible when combined with microbubble ultrasound contrast agents. These agents may also make thermal coagulation possible at lower acoustic energies.12
Clinical trials of extracorporeal low-frequency transcranial ultrasound for brain ischemia with the assistance of a tissue plasminogen activator have shown promise in the recanalization of thrombosed vessels. 13
Prostate tumors. Prostate cancer accounted for 33% of newly presenting cancers in U.S. men in 2006, with 88% of the newly diagnosed tumors being confined to the prostate.14 Many prostate tumors are amenable to local treatments such as prostatectomy and local radiotherapy. These strategies are effective in terms of disease control, but they are also associated with side effects, including urinary incontinence and impotence.
Side effects are kept to a minimum with MRgFUS. An endorectal focused ultrasound transducer creates well-defined areas of tumor necrosis, sparing adjacent normal prostatic tissue. Catheter-based (intra-urethral) focused ultrasound devices with endorectal MRI coils have also been developed. Results from ongoing clinical trials indicate that focused ultrasound treatment of prostate cancer is now challenging radiation therapy. Treatment is particularly effective in patients with a Gleason score of six or less, and when the PSA is 10 ng/mL or lower.15
Bone metastases. Thirty percent of cancer sufferers develop bone metastases, the majority of which result in significant pain.16 Existing methods of pain palliation include radiotherapy, analgesia, hormone therapy, and RF ablation.
MRgFUS has been identified as a safe, effective, and repeatable noninvasive means of treating painful bone metastases. The absorption of acoustic energy is 50 times higher in bone than in soft tissues, allowing heating to be localized at the cortex. Metastatic bone pain is thought to be caused by the involvement of periosteal nerves, and focused ablation of the periosteum causes localized permanent denervation (Figure 5).
Results from a multicenter study showed that the mean VAS score (a robust pain measurement tool) in patients with painful bone metastases dropped from 5.9 (pretreatment) to 1.8 (three months post-treatment).16 The effects are usually seen within days of the treatment. The relief from pain may be so dramatic that patients are advised to avoid excessively demanding activities in order to reduce the risk of developing a pathological fracture.
MRgFUS treatment of facet joint-related pain, by accurate ablation of facet joint innervation, is currently being evaluated.
RISKS AND REWARDS
MRgFUS is an extremely safe treatment modality when used correctly and appropriately.
Most treatments are single-stage procedures, carried out under light sedation on an outpatient basis, with consequent benefits regarding treatment cost and patient experience.
Rare complications include skin burns and damage to the bowel and nerves. Careful patient selection, as well as thermal mapping, presonication planning, and the avoidance of nerves, bowel, and cutaneous scars should virtually eliminate the occurrence of these complications.
It is likely that indications and uses for MRgFUS will continue to grow. The long-term aim is that this safe and effective nonionizing treatment modality will replace many conventional invasive procedures in the organs described above. Future work will also exploit the inherent local heating capabilities of MRgFUS to activate drugs that have been delivered systemically, a strategy that will enable organ-specific treatment. This is a particularly exciting development that has huge implications for reducing or eliminating the generalized side effects that are frequently seen with chemotherapy.
The ExAblate MRgFUS system (InSightec) has CE and U.S. Food and Drug Administration approval for the treatment of uterine fibroids, as well as CE approval for pain palliation of bone metastases. All other applications of MRgFUS are at the clinical trials stage and are not yet widely available.
Articles in this issue
almost 15 years ago
Intramedullary Spinal Sarcoidosis Myelopathyover 15 years ago
Marilyn Monroe's chest x-rays bring big moneyover 15 years ago
PET agent unlocks door to Alzheimer’s diseaseover 15 years ago
Endovascular embolization stops nosebleedsover 15 years ago
PET with CTC detects polyps well, requires no cleansingover 15 years ago
US identifies patients at higher stroke riskover 15 years ago
Algorithm lowers number of CTs for some strokesover 15 years ago
Letters to the Editorover 15 years ago
Vertebroplasty study adds weight, but not final wordover 15 years ago
Ultrasmall technology promises big boostNewsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.