SuperSonic promotes novel form of elastography
SuperSonic Imagine returned to the RSNA exhibit floor with FDA clearance to market its Aixplorer ultrasound system in the U.S.
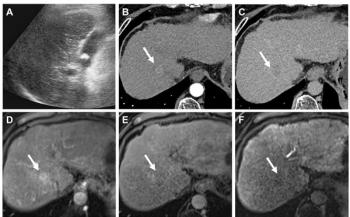
SuperSonic Imagine returned to the RSNA exhibit floor with FDA clearance to market its Aixplorer ultrasound system in the U.S. The French company last year unveiled its software-based system along with its ShearWave Elastography technique as the means for differentiating benign and malignant lesions of the breast. Although the digital measurements that define the stiffness of these tissues can be displayed and interpreted on Aixplorer systems sold outside the U.S., the FDA-cleared version shows only B-mode color-coded images. These images show the relative stiffness or elasticity of tissue, which may help physicians distinguish among healthy tissue and benign and malignant tumors in the breast. The company is now developing applications for the thyroid and abdomen, which will define cancer as well as other diseases.
Aixplorer creates these images using algorithms developed for video games and multicore processors. The company's ShearWave Elastography differs from conventional, or strain, elastography, which relies on manual compression by the operator and is, therefore, dependent on operator skill. Instead, ShearWave elastography is operator-independent, using both ultrasound waves and shear waves to assess tissue stiffness.
Newsletter
Stay at the forefront of radiology with the Diagnostic Imaging newsletter, delivering the latest news, clinical insights, and imaging advancements for today’s radiologists.