Carestream Receives FDA Clearance for Digital Tomosynthesis
FDA clearance extends digital tomosynthesis to Carestream customers.
Carestream received 510(k) clearance from the U.S. Food and Drug Administration for its digital tomosynthesis (DT) functionality on Tuesday.
As an upgradable option on the existing Carestream DRX-Evolution Plus System, the DT technology can simplify workflow and reduce exam time, according to a company press release. It will now be available to customers for purchase, and it can be paired with Carestream’s new ImageView Software that is powered by the company’s Eclipse image processing engine.
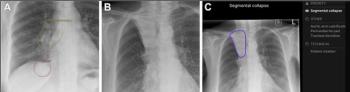
Similar to CT scan, DT can produce cross-sectional images of any organ. However, it uses a single sweep to capture a series of low-dose X-ray images. This functionality will increase visibility for radiologists in time-critical situations, said Sarah Verna, Carestream’s worldwide marketing manager for global X-ray solutions, in the release.
“In trauma centers, CT rooms are always overbooked, and there’s always a wait,” she said. “Now, a doctor can further examine any body part, including the chest or lungs, with digital tomosynthesis because it generates many image slices, which helps sharpen diagnosis and make medical treatment more efficient.”
The intent, she said, is to use DT in collaboration with CT – not to replace it – while streamlining workflow in hospital systems and practices.