Study Finds AI More Effective Than Sonographer Interpretation of Cardiac Function on Echocardiograms
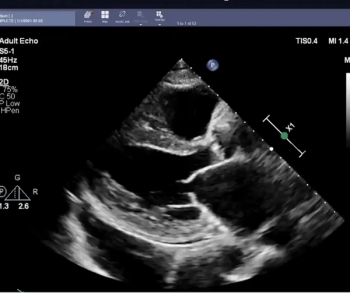
Based on a review of 3,495 echocardiographic studies to evaluate left ventricular ejection fraction (LVEF), researchers found that cardiologists changed initial artificial intelligence (AI) assessment 16.8 percent of the time and initial sonographer assessment 27.2 percent of the time.